Imaging in Alzheimer‘s
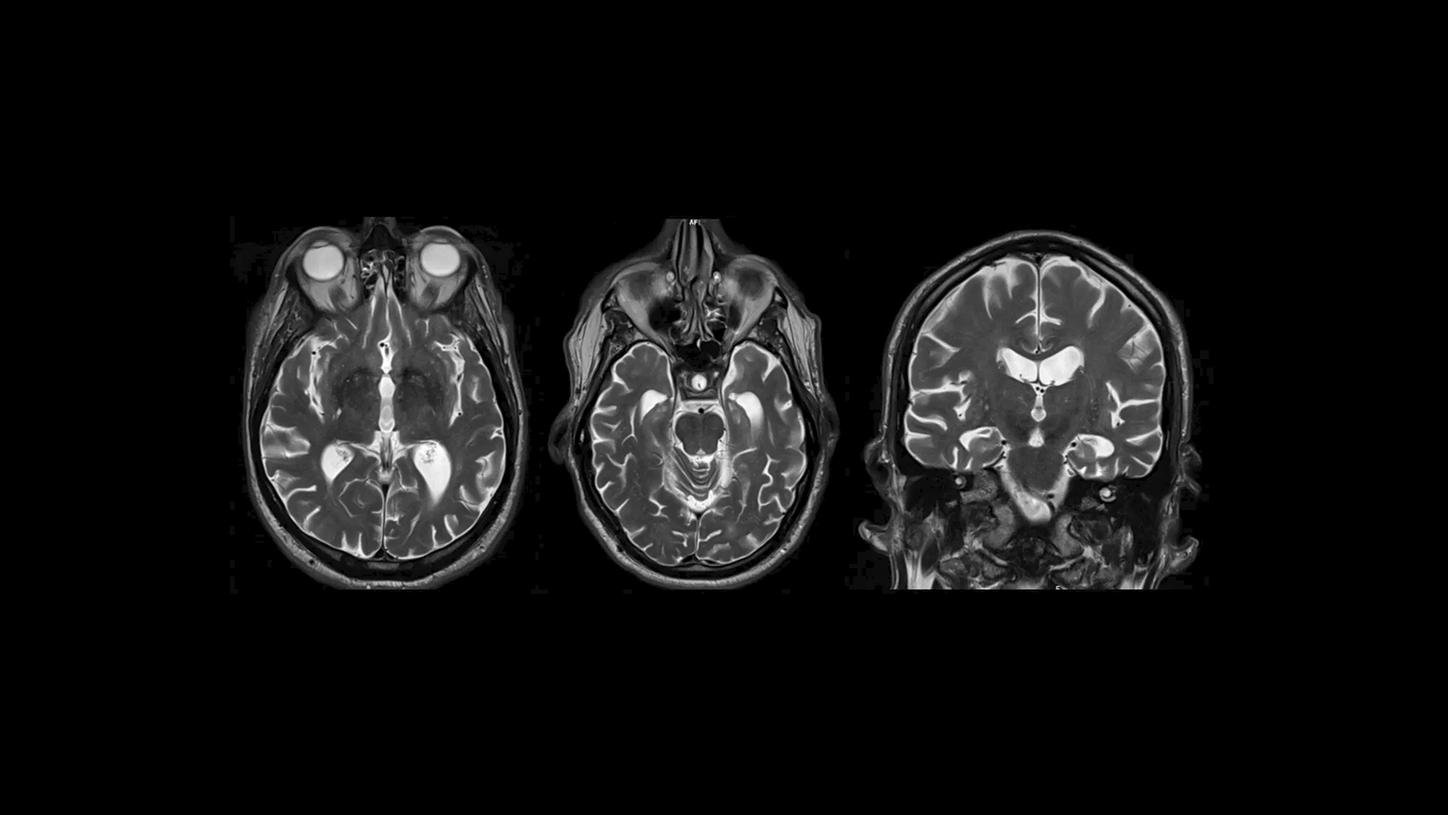
On T2 MR imaging, water and cerebrospinal fluid appear as bright areas. This is due to their high signal intensity.
For Alzheimer’s patients, timely access to MRI can change everything. AI can support scans that are up to 50 percent faster—so more patients get imaged sooner, helping treatment decisions and follow-up.
André Hartung, President Diagnostic Imaging, Siemens Healthineers
A PET/MR image of a patient with Alzheimer’s disease.²
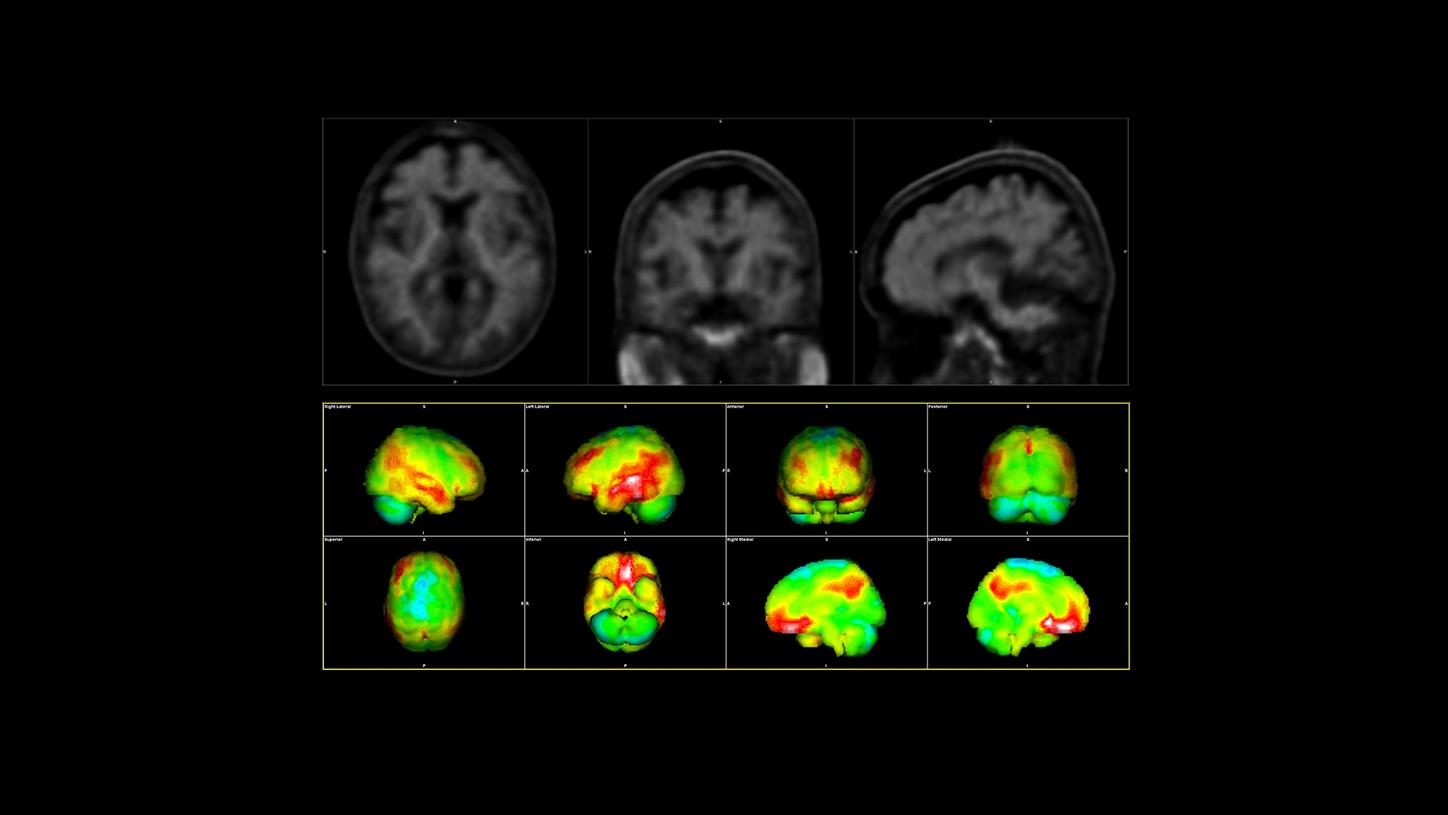
A typical amyloid PET scan of a patient with Alzheimer’s disease: The red areas are the affected regions in the brain.³
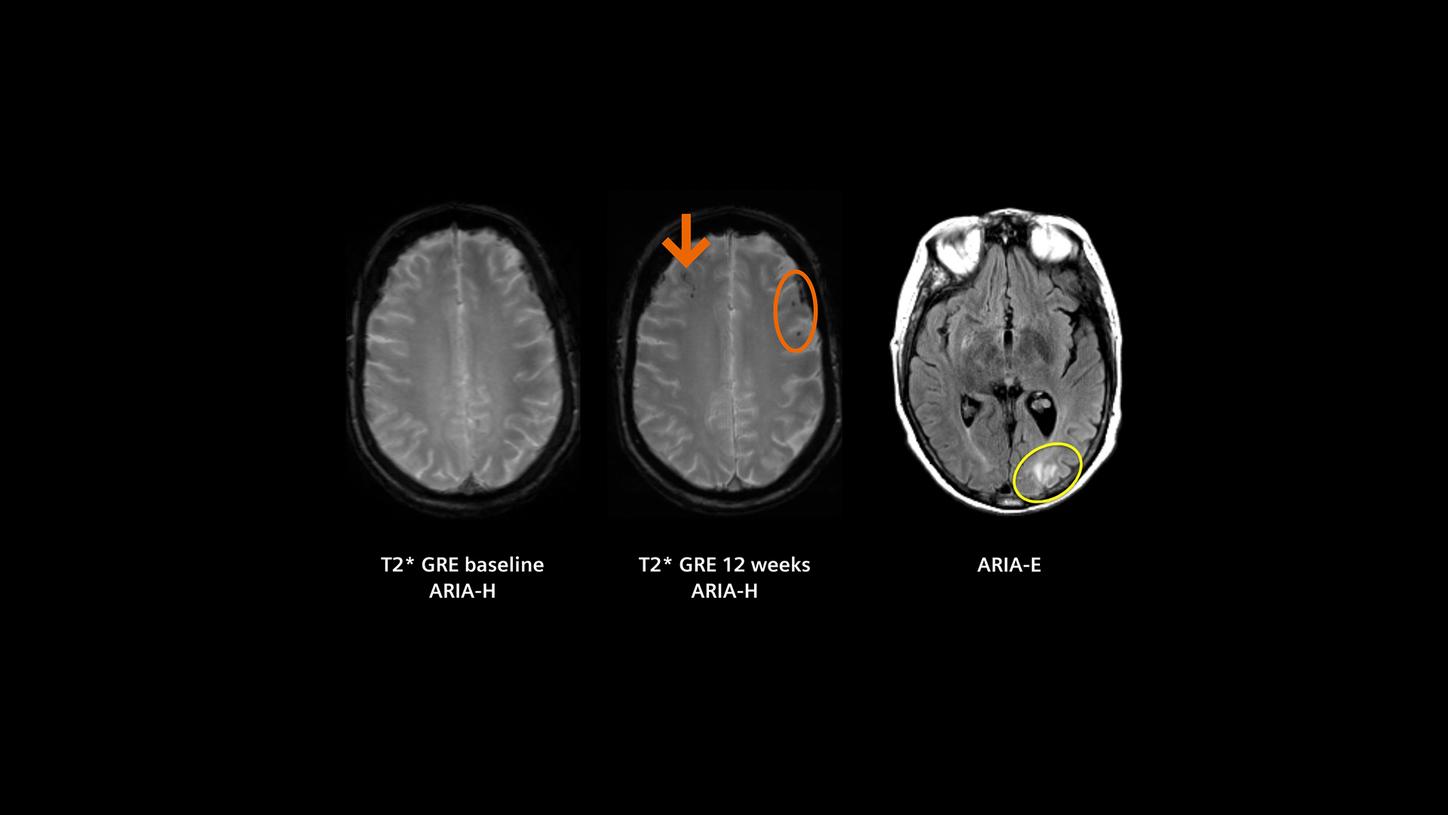
MR images of amyloid-related imaging abnormalities (ARIA-H and ARIA-E). These are potential adverse side effects of the new disease-modifying therapies in Alzheimer’s disease.
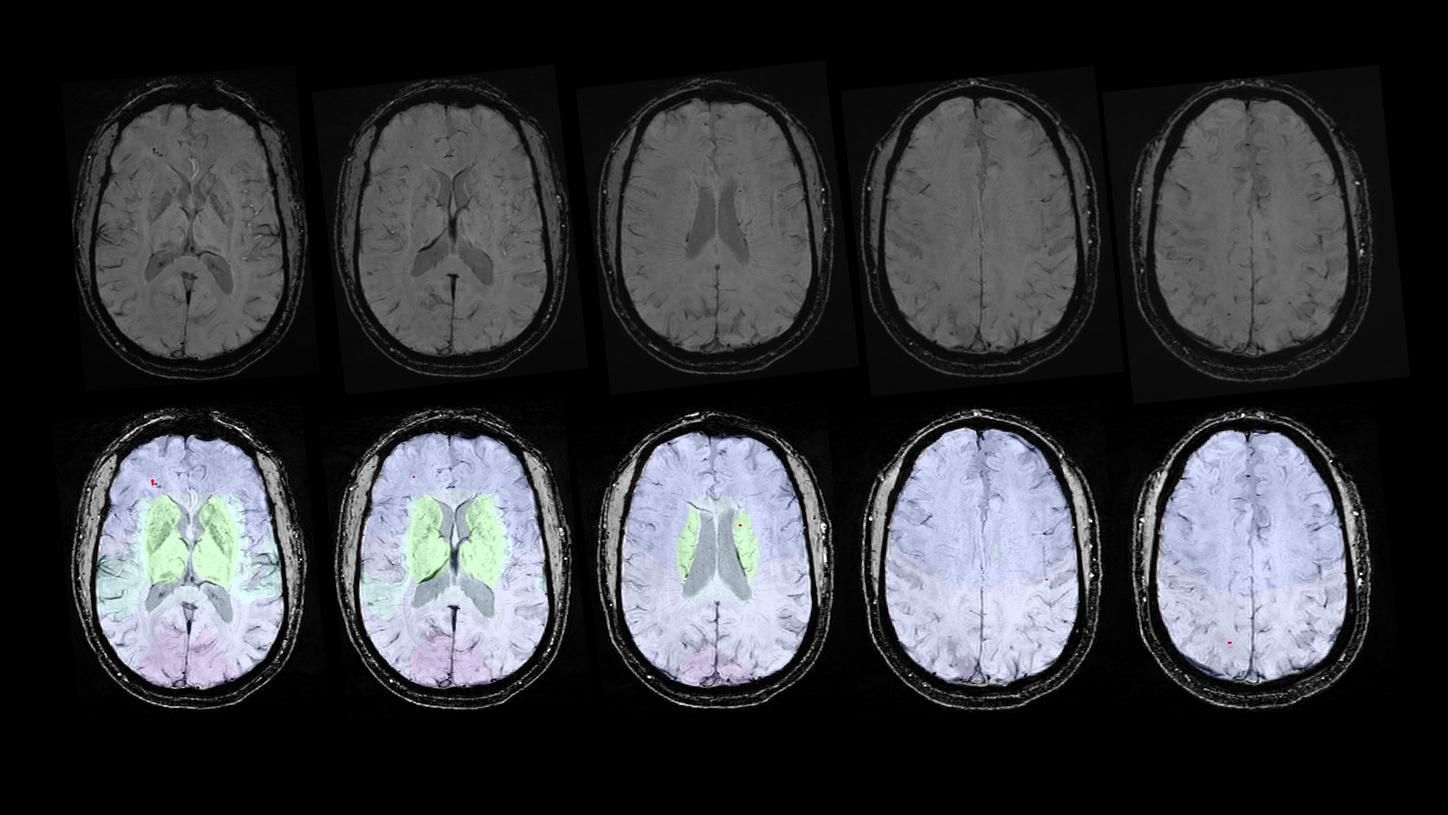
Identifying cerebral microbleeds with the help of AI (prototype).
1 Disease-modifying therapies (DMTs) in Alzheimer’s disease come with risks. Please refer to the specific therapy prescribing information for side effects, contraindications, and risks.
2 Acquired on Biograph mMR | Study-ID: 4aaaa0025 | Image courtesy of University Hospital Plzen, Czech Republic.
Images from Biograph mMR are provided to illustrate typical PET/MR imaging capabilities and are not specific to Biograph One. Biograph One is pending 510(k) clearance and is not yet commercially available in the USA. Its future availability cannot be guaranteed.3 Data courtesy of Wentworth-Douglass Hospital, Dover, New Hampshire, USA.
4 This application is intended for research purposes only and must not be used for patient-individual diagnostic use. The application mentioned may not be available in all countries. Please contact your Siemens Healthineers representative for more information about this research application as well as its availability in your country.
References[1] Furtner J, Prayer D. Neuroimaging in dementia. Wien Med Wochenschr. 2021;171(11–12):274–81.
[2] Molinder A, Ziegelitz D, Maier SE, Eckerström C. Validity and reliability of the medial temporal lobe atrophy scale in a memory clinic population. BMC Neurol. 2021;21(1):289.
[3] Shi Q, Hou J, Peng X, Xu Z, Wang Y, Cao D. Magnetic resonance imaging analysis for Alzheimer's disease diagnosis using artificial intelligence: Methods, challenges, and opportunities. Ageing Res Rev. 2026;113:102943. Epub 2025 Nov 19.
[4] U.S. Food & Drug Administration. FDA to recommend additional, earlier MRI monitoring for patients with Alzheimer’s disease taking Leqembi (lecanemab). Current as of August 28, 2025. Accessed January 1, 2026. Available from: https://www.fda.gov/drugs/drug-safety-and-availability/fda-recommend-additional-earlier-mri-monitoring-patients-alzheimers-disease-taking-leqembi-lecanemab











