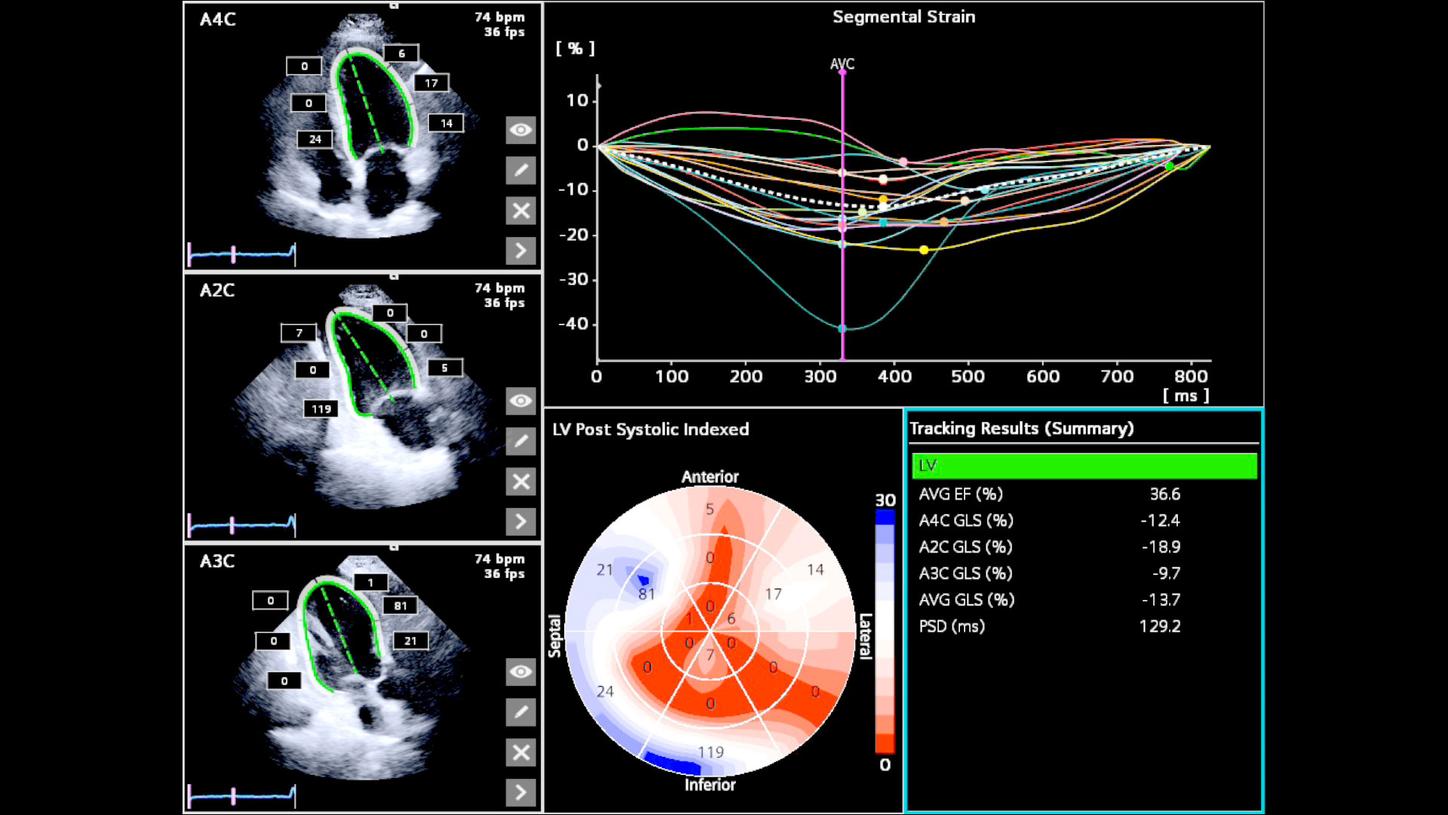
In cardio-oncology, small changes matter. “When we need to monitor patients who receive cardiotoxic therapies, we face two main challenges,” explains Lopez-Fernandez. “We must schedule our echo timely, the correct day to minimize the risk of cancer treatment delays, but we also need to be very reproducible.” Changes in GLS provide an early signal that the heart may be under stress. These findings lead to closer monitoring and joint assessment by the cardiology and oncology teams, allowing the implementation of cardioprotective strategies while maintaining cancer treatment.
Further changes in cardiac function — in particular, a significant decline in left ventricular ejection fraction (LVEF) — also guide decisions on further intervention. “When we get these numbers, we can make appropriate clinical decisions,” says Oliver Higuera Gómez, MD, oncologist at La Paz University Hospital in Madrid. Cardio-oncology teams rely on these cardiac measurements to assess risk, initiate cardioprotection or heart failure therapy when needed, and support the continuation of cancer treatment. This is where reproducibility becomes essential.