Explore our comprehensive portfolio that addresses the complete care continuum for many of the world’s most threatening diseases.
Business partners
Conferences & events
Discover the stories of pioneers pushing the boundaries of what’s possible in healthcare to help improve people’s lives around the world.
Patient-centered innovation has been and always will be at the core of our company. Dive deeper into our fascinating technologies.
We pioneer breakthroughs in healthcare. For everyone. Everywhere. Sustainably. Learn more about our purpose, our innovation leadership, our culture, and our strategy.
Our responsibility
Our career opportunities
Home
- Press center
- Press releases
- FAST III Study Validates Less-Invasive Method for Coronary Revascularization Guidance as Safe and Effective Alternative
FAST III Study Validates Less-Invasive Method for Coronary Revascularization Guidance as Safe and Effective Alternative
- FAST III started out five years ago to test whether vessel fractional flow reserve (vFFR)-guided PCI leads to similar therapeutic outcomes as interventions guided by pressure-wire based FFR
- Positive results validate software Caas vFFR as a safe and effective alternative for guiding revascularization
- Paves the way for further clinical adoption of less-invasive physiology testing for patients with suspected coronary stenoses
- Study sponsored by the European Cardiovascular Research Institute (ECRI) and co-funded by industry partners Siemens Healthineers and Pie Medical Imaging
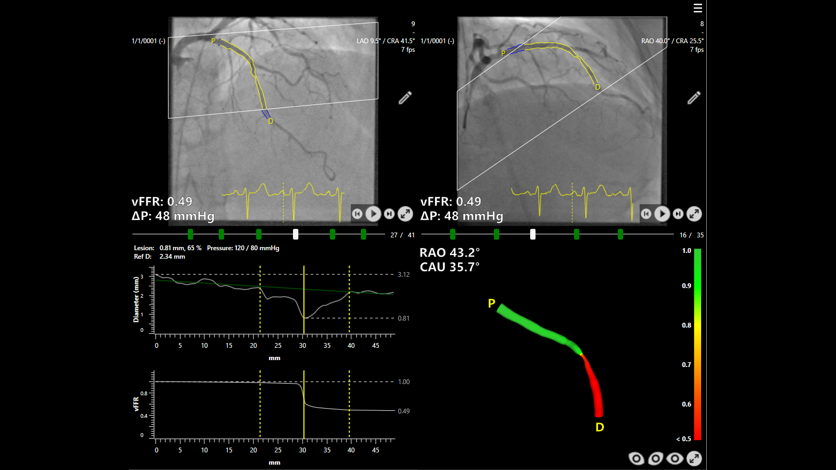
Today the results of the FAST III study, co-funded by Siemens Healthineers and Pie Medical Imaging, were presented at the Congress of the American College of Cardiology (ACC) and published in The New England Journal of Medicine (Angiography-Based Physiology to Guide Coronary Revascularization | New England Journal of Medicine). The results confirm the safety and efficacy of Caas vFFR – a software-based method developed by Pie Medical Imaging that calculates pressure drop and vessel fractional flow reserve from two angiographic images – as an alternative to conventional pressure wire-based physiology for guiding revascularization strategy in patients with intermediate coronary artery stenosis. This marks the end of the multicenter, randomized-controlled, open-label, non-inferiority study investigating the outcome of therapy for coronary artery disease after fractional flow reserve (FFR) has been determined using different methods. A total of 2,235 patients in 37 hospitals across seven European countries took part in the study, randomly selected for either the vFFR- or FFR-guided approach. FAST III marks the last important step in a ten-year evidence generation journey towards real-world outcome data.
“We are extremely proud having finalized this important landmark trial,” said Dr. Joost Daemen, chief investigator of FAST III and cardiologist in the Thoraxcenter at the Erasmus University Medical Center in Rotterdam, Netherlands. “The use of vFFR eliminates the need for guiding catheters, invasive coronary artery instrumentation and hyperemic agents with inherent risks and patient discomfort. This may streamline procedures, allows the use of physiology in centers in which only diagnostic coronary angiography is performed and may help to guide heart team decision making. FAST III has the potential to reset the discussion about European guidelines for the management of chronic coronary syndrome by providing robust evidence on the safety and efficacy of angiography-based physiology as a valid alternative to conventional pressure wire-based physiology.”
Coronary stenoses are extremely common and clinically relevant as they sit at the center of coronary artery disease (CAD), the leading cause of death globally. FFR assesses how much coronary artery stenosis limits blood flow, with evidence showing that lesions causing less than a 20 percent flow reduction generally do not require treatment. Traditionally, FFR is measured by inserting a pressure wire into the coronary artery and inducing hyperemia with adenosine, a process that can cause the patients discomfort such as restlessness, chest pain, and shortness of breath. The FAST III study had evaluated whether percutaneous coronary intervention (PCI) guided by Caas vFFR can match the outcomes of the conventional invasive, pressure wire approach. This newer Caas vFFR technology offers a less invasive alternative by eliminating the need for both a pressure wire and hyperemic agents.
“The results of the FAST III study mark a significant milestone for cardiovascular care in Europe and for our partnership with Pie Medical Imaging,” said Doris Pommi, head of Cardiovascular Care at Siemens Healthineers. “Its success shows how much we can achieve when we join forces and combine scientific excellence, clinical experience, and industrial innovation. The patient impact of the study is substantial - we are looking at safer, faster, less uncomfortable coronary assessments with potentially lower costs and wider access to physiologic lesion evaluation.”
René Guillaume, recently retired CEO of Pie Medical Imaging said: “As one of the two grant providers, Pie Medical Imaging, together with Siemens Healthineers, highlights the importance of translating advanced technologies into clinical value for healthcare professionals and patients. Unbiased results from clinical outcome studies are crucial to demonstrating the reliability and accuracy of the CAAS vFFR software.”
The leading clinical centers involved in FAST III included but were not limited to Erasmus University Medical Center (The Netherlands), John Radcliffe Hospital Oxford (UK), Mater Private Hospital Dublin (Ireland), University Hospital of Verona (Italy), University Hospital Valladolid (Spain), Charité Berlin (Germany), and University Hospital Lille (France). The trial was successfully executed through Cardialysis, a leading European cardiovascular research organization based in Rotterdam, Netherlands.
Further information about vFFR can be found here: https://www.siemens-healthineers.com/angio/options-and-upgrades/clinical-software-applications/angio-derived-ffr-measurement
Image Gallery

CAAS vFFR software by Pie Medical Imaging, as used in the FAST III study.
Download image

Dr. Joost Daemen, chief investigator of FAST III and cardiologist in the Thoraxcenter at the Erasmus University Medical Center in Rotterdam, Netherlands: “We are extremely proud having finalized this important landmark trial. The use of vFFR eliminates the need for guiding catheters, invasive coronary artery instrumentation and hyperemic agents with inherent risks and patient discomfort. This may streamline procedures, allows the use of physiology in centers in which only diagnostic coronary angiography is performed and may help to guide heart team decision making.”
Download image
Download this press release
(pdf, 287.53 KB)Contact

Kathrin Palder
Innovation, Artificial Intelligence, Advanced Therapies









