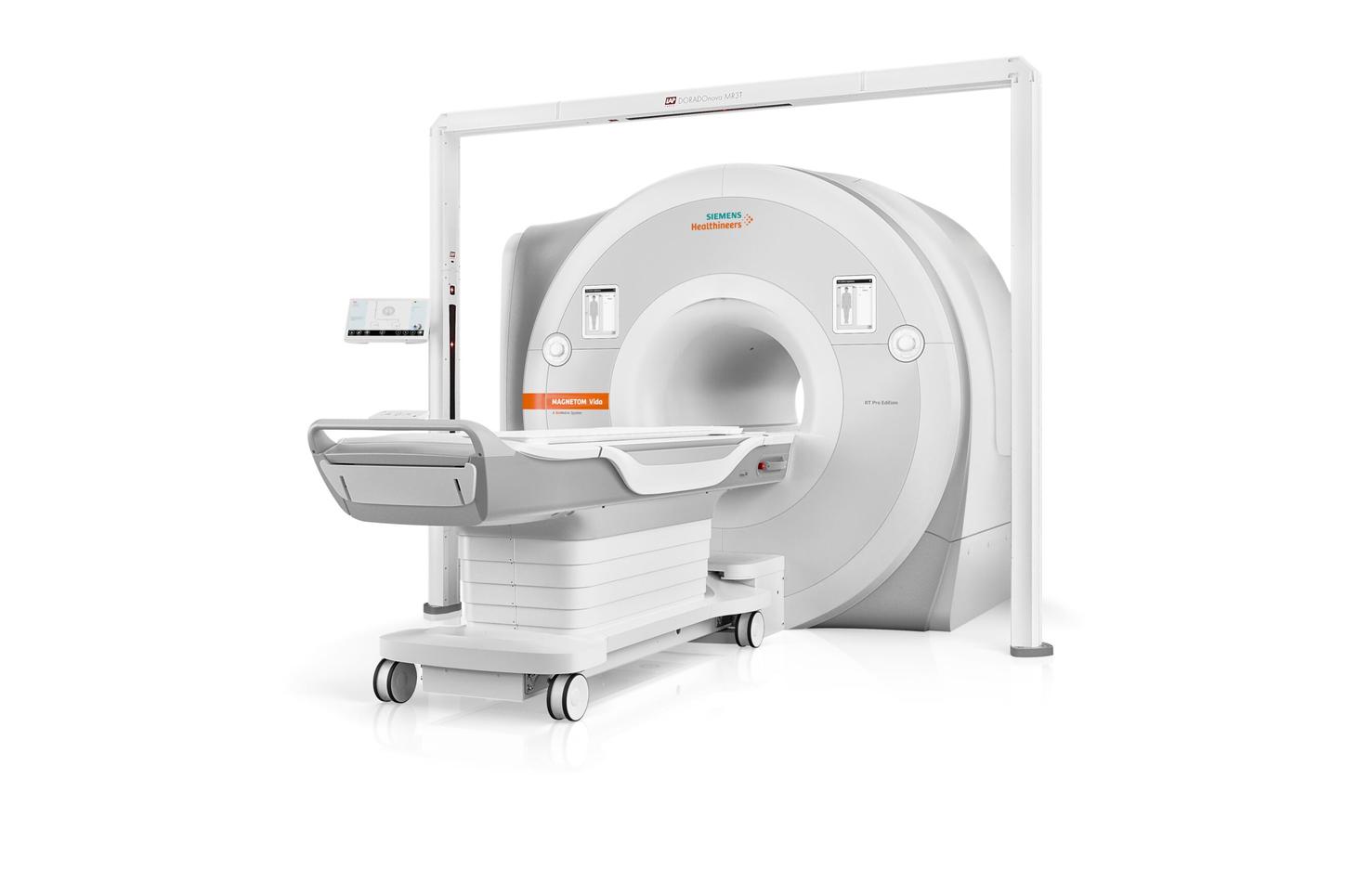
RT Pro Edition for MAGNETOM Sola and VidaBringing the power of MRI to RT
The RT Pro Edition for MAGNETOM Sola and MAGNETOM Vida addresses the challenges of patient variability for RT treatment planning.
MAGNETOM Sola and MAGNETOM Vida deliver excellent soft-tissue contrast as well as imaging that can yield information for physicians regarding tumor extent and functional parameters.
Efficiently integrating this additional modality into existing processes can add substantial value to radiotherapy and help advance your institution’s clinical capabilities.

Benefits
BioMatrix Technology, available on MAGNETOM Sola and MAGNETOM Vida, truly personalizes MRI and increases precise, reproducible imaging for more efficient RT planning, monitoring, and follow-up. BioMatrix consists of three key technologies: BioMatrix Sensors are respiratory sensors that automatically display a patient’s breathing curve and actively trigger acquisition sequences at the right point in time. BioMatrix Tuners improve the correlation of DWI to corresponding anatomical scans. With BioMatrix Interfaces, the BioMatrix dockable table with eDrive helps you move your patient effortlessly to and from the scanner, and docks automatically with one touch.
In order to provide precise and reproducible patient positioning, RT Pro Edition for MAGNETOM Sola and Vida offers dedicated hardware for high-quality MR imaging in treatment position. To fully utilize MR imaging capabilities for RT treatment planning, we’ve partnered with leading companies in the RT market (LAP,1 Qfix,1 CIVCO,1 and Orfit1) to provide positioning and patient marking equipment which is compatible with both scanners. Furthermore, MAGNETOM Sola and MAGNETOM Vida come with a wide range of coils, enabling easy and fast patient positioning as well as precise image quality in treatment position. Thanks to an ultra-high coil-element density, new BioMatrix and UltraFlex coils provide increased SNR and enable high acceleration factors that can save considerable time.
Adding MRI into RT workflows can be challenging, especially for those who have less experience in MR imaging. myExam RT Assist4 and syngo.via RT Image Suite help users to incorporate MRI into RT by providing workflow guidance – from image acquisition to postprocessing and contouring.2 MR-based Synthetic CT, a feature of syngo.via RT Image Suite, enables users to implement an MR-only radiotherapy workflow.3 In order to perform precise target delineation, 4D MRI – RT Respiratory Self-Gating helps you caption organ motion in abdomen and thorax with automatic respiratory phase sorting during the entire free-breathing acquisition.
Evidence
Technical Details
Learn more about the RT Pro Edition for MAGNETOM Vida and MAGNETOM Sola