RadiopharmaDelivering a broad portfolio of PET radiopharmaceuticals to advance patient outcomes
What's new?
Who we are
Radiopharma
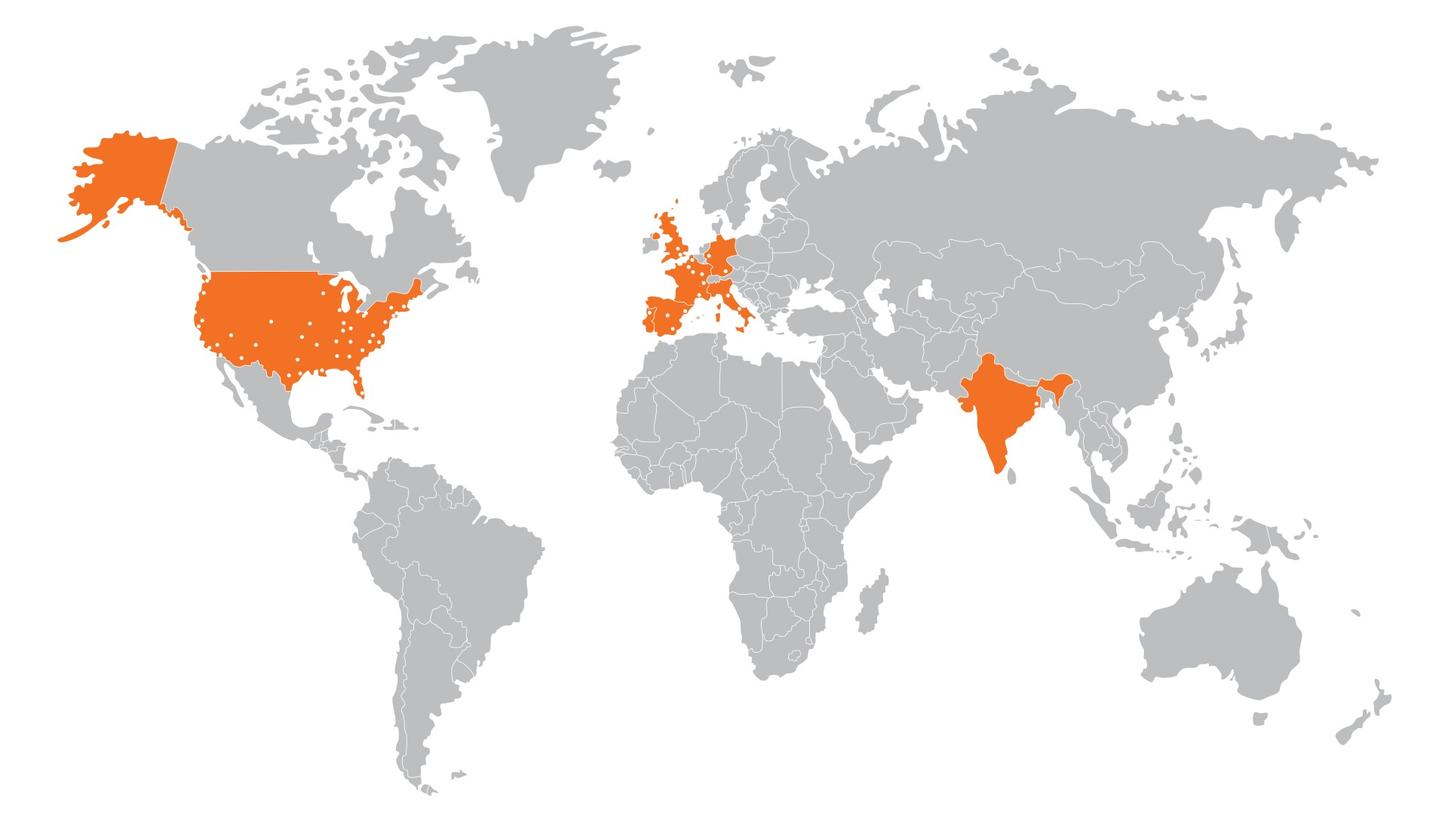
As part of Siemens Healthineers, our Radiopharma business is committed to developing, manufacturing, and delivering positron emission tomography (PET) radiopharmaceuticals via the largest network of PET manufacturing facilities globally. We manufacture and deliver more than 2,100,000 doses1 to more than 3,000 imaging centers1 each year. In the United States alone, we deliver at least one product to 92% of PET imaging centers.2
Contract Development and Manufacturing Organization of Choice
As a leading PET radiopharmaceutical manufacturing and service provider across the US and Europe, we offer scalable, efficient solutions to third-party drug developers as a preferred contract development and manufacturing organization (CDMO). We support third-party drug developers in bringing innovative tracers to market for personalized medicine and theranostics, while tailoring our services to meet regional needs and ensuring consistent, high-quality solutions globally.
And as a key part of Siemens Healthineers Molecular Imaging, we have a direct line into front-line research and development in the world of molecular imaging.
Our locations
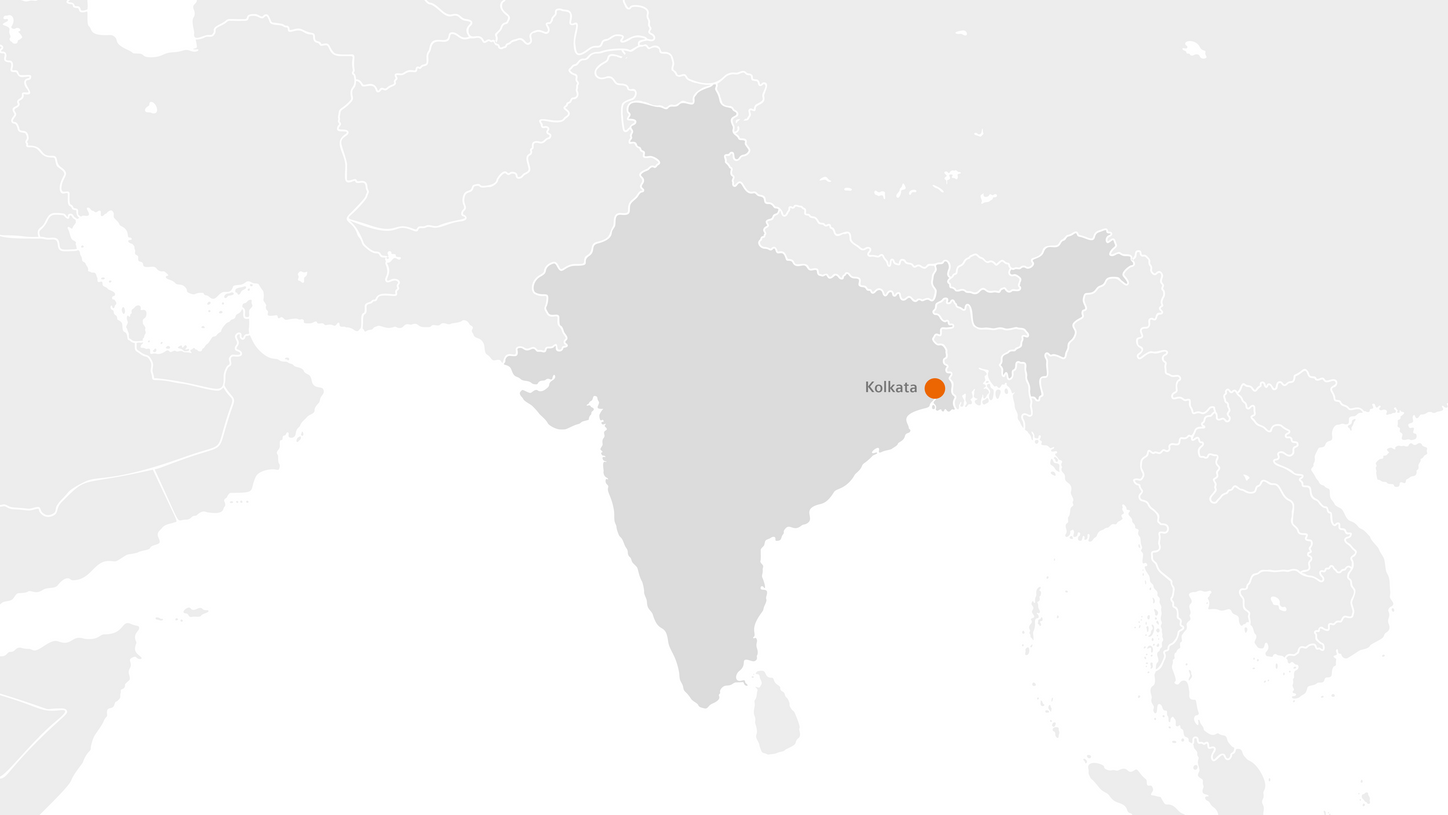
India
Radiopharma in Kolkata is a trusted provider of high-quality PET radiopharmaceuticals across the region. Strategically located, it serves an extensive network across northeastern India—including Guwahati, Patna, Bhubaneswar, Ranchi, Agartala, and Imphal—and extends its reach internationally to Nepal.
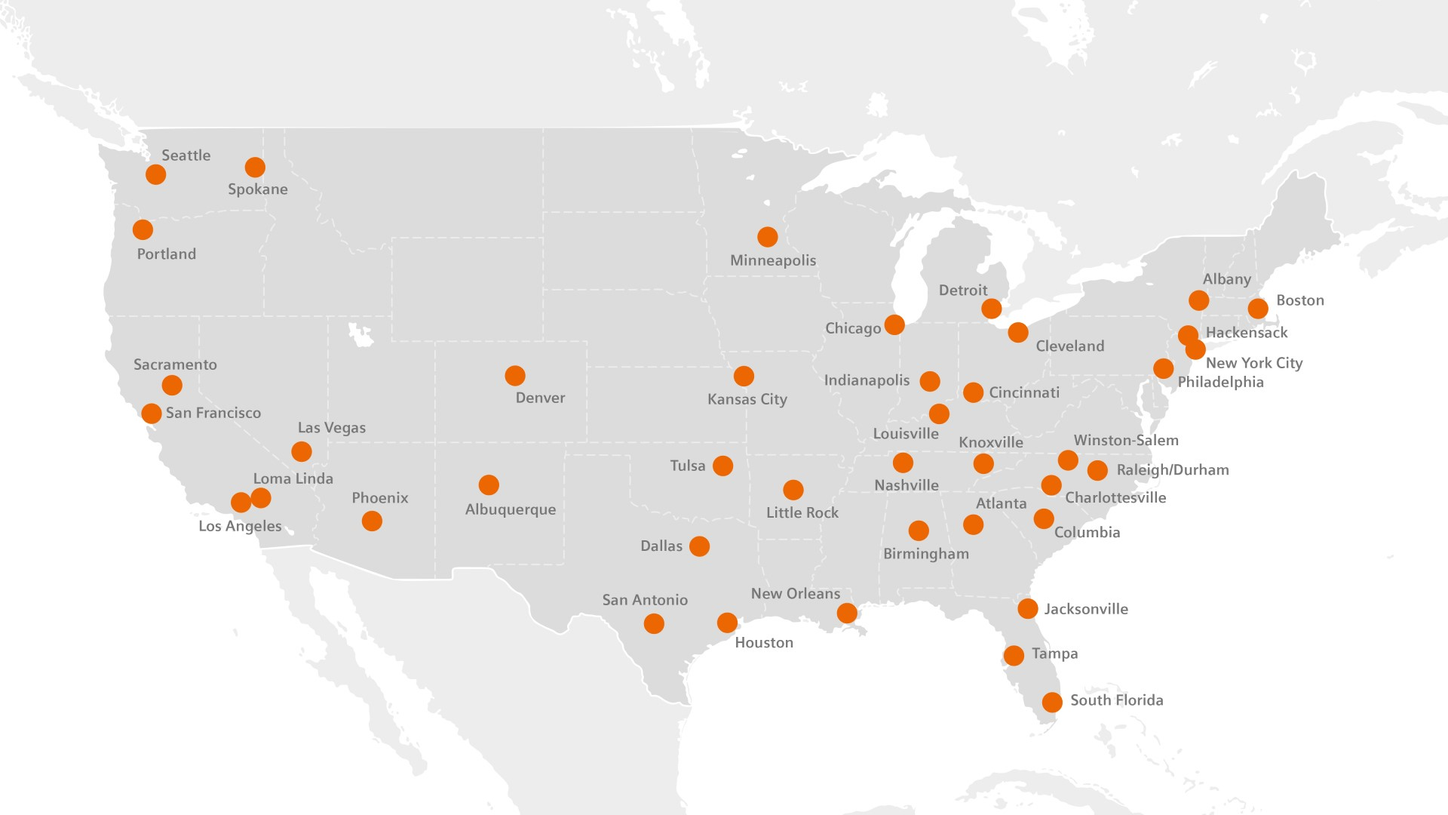
USA
Onboard with the Radiopharma business of Siemens Healthineers
Is Radiopharma on contract with any GPOs?
Yes, we are on contract with the top GPOs in the nation. Activate through the GPO portal and follow the steps below.
What if I'm not a Radiopharma customer?
New customers will complete a simple 3-step onboarding process:
- Sign the member agreement
Direct inquiries to contracts@petnetsolutions.com - Send onboarding form and RAM license to Radiopharma
Required for each ship-to site. Allow up to 5 days processing time - Receive customer welcome
Receive customer ID and instructions for ordering & scheduling doses
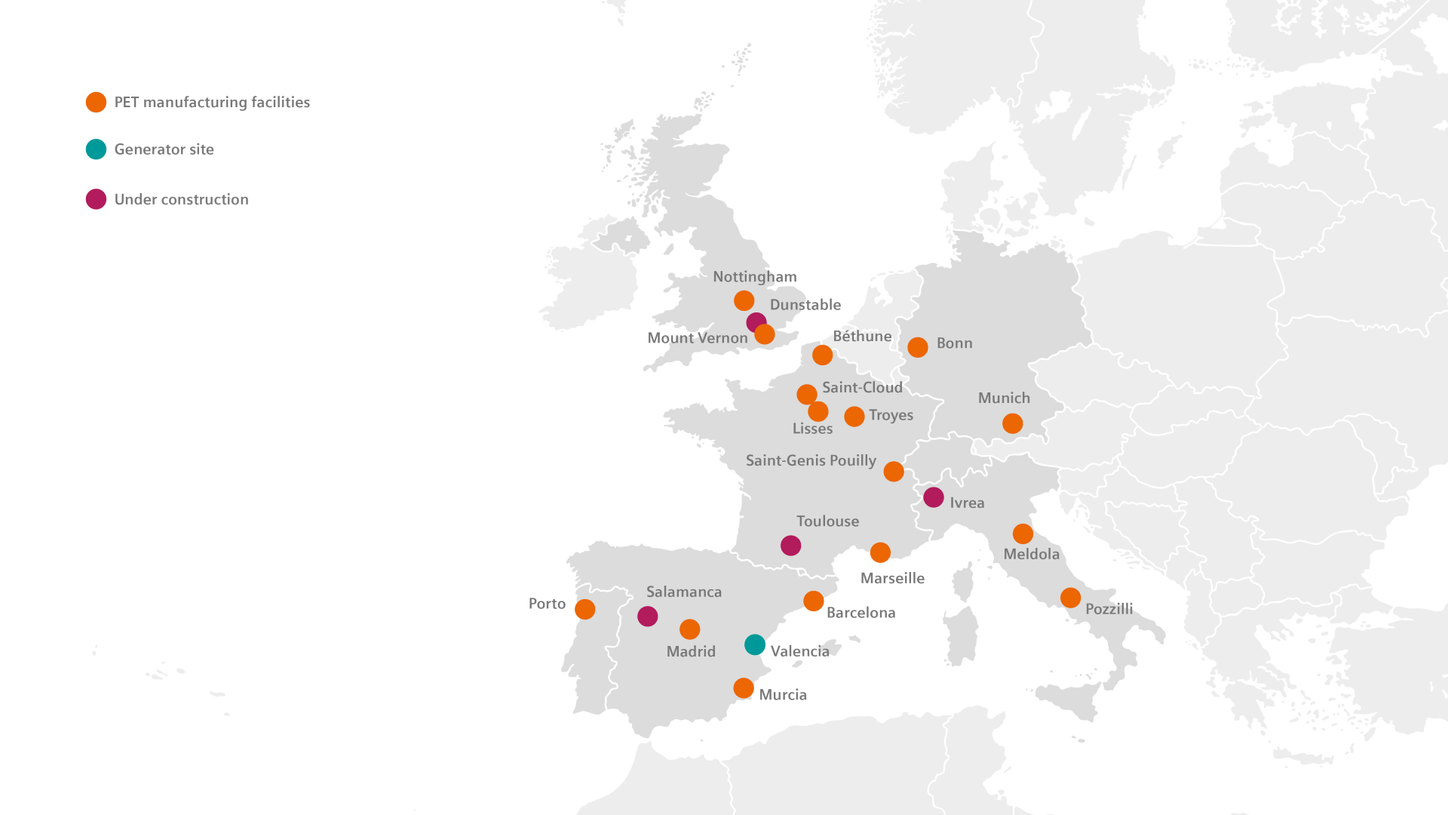
Europe
As of December 2, 2024, Siemens Healthineers has completed the acquisition of Advanced Accelerator Applications Molecular Imaging. This acquisition expands Siemens Healthineers' global manufacturing footprint to a total of 60 facilities, including new sites in France, Spain, Portugal, Italy, and Germany.

India
Radiopharma in Kolkata is a trusted provider of high-quality PET radiopharmaceuticals across the region. Strategically located, it serves an extensive network across northeastern India—including Guwahati, Patna, Bhubaneswar, Ranchi, Agartala, and Imphal—and extends its reach internationally to Nepal.

USA
Onboard with the Radiopharma business of Siemens Healthineers
Is Radiopharma on contract with any GPOs?
Yes, we are on contract with the top GPOs in the nation. Activate through the GPO portal and follow the steps below.
What if I'm not a Radiopharma customer?
New customers will complete a simple 3-step onboarding process:
- Sign the member agreement
Direct inquiries to contracts@petnetsolutions.com - Send onboarding form and RAM license to Radiopharma
Required for each ship-to site. Allow up to 5 days processing time - Receive customer welcome
Receive customer ID and instructions for ordering & scheduling doses

Europe
As of December 2, 2024, Siemens Healthineers has completed the acquisition of Advanced Accelerator Applications Molecular Imaging. This acquisition expands Siemens Healthineers' global manufacturing footprint to a total of 60 facilities, including new sites in France, Spain, Portugal, Italy, and Germany.

India
Radiopharma in Kolkata is a trusted provider of high-quality PET radiopharmaceuticals across the region. Strategically located, it serves an extensive network across northeastern India—including Guwahati, Patna, Bhubaneswar, Ranchi, Agartala, and Imphal—and extends its reach internationally to Nepal.




Clinical trial support
We are committed to advancing healthcare through strategic partnerships that enable participation in clinical trials across the globe. In collaboration with leading pharmaceutical companies, we support the development and evaluation of novel radiopharmaceutical agents and facilitate the submission of regulatory applications. Beyond providing access to radiopharmaceuticals, we offer comprehensive training and operational support to ensure trial sites are fully equipped to conduct high-quality research and contribute to meaningful clinical outcomes.
Our portfolio
Broadest portfolio of PET radiopharmaceuticals
Whether your focus is on clinical care or investigational research, we offer the broadest portfolio of PET radiopharmaceuticals manufactured and delivered from a single supplier:
Case studies

Enhancing healthcare excellence:
Improving patient access and driving business growth with PETNET Solutions
As the patient demand for PET imaging grows, imaging centers need a reliable PET radiopharmaceutical partner to support them. Read the case study to learn how Sentara Health improves patient access and drives business growth with PETNET Solutions.

Enhancing healthcare excellence:
Expert support in action
Through a long-standing partnership with PETNET Solutions, RAYUS Radiology has expanded its business by leveraging expert support and a comprehensive PET tracer portfolio to improve patient outcomes. Read the case study to see how this collaboration is making a difference.

Enhancing healthcare excellence:
Improving patient access and driving business growth with PETNET Solutions
As the patient demand for PET imaging grows, imaging centers need a reliable PET radiopharmaceutical partner to support them. Read the case study to learn how Sentara Health improves patient access and drives business growth with PETNET Solutions.

Enhancing healthcare excellence:
Expert support in action
Through a long-standing partnership with PETNET Solutions, RAYUS Radiology has expanded its business by leveraging expert support and a comprehensive PET tracer portfolio to improve patient outcomes. Read the case study to see how this collaboration is making a difference.

Enhancing healthcare excellence:
Improving patient access and driving business growth with PETNET Solutions
As the patient demand for PET imaging grows, imaging centers need a reliable PET radiopharmaceutical partner to support them. Read the case study to learn how Sentara Health improves patient access and drives business growth with PETNET Solutions.


Expert support
*Not available in all countries
Our knowledgeable staff averaging more than 20 years of industry experience supports your imaging practice and encourages growth. You can rely on our high performance customer care, pharmacy, and sales teams for ordering and scheduling support, market development, and strategic account engagement.
Seamless delivery
More than 16 million doses delivered since 2011
We maintain a dose fulfilment rate of >99% through our integrated network of radiopharmacies. We know it can be challenging to maintain patient schedules, minimize wait times, and provide timely results to referring physicians without PET tracer delivery you can count on.
Pomohli vám tieto informácie?
Ďakujeme
Based on 2020 IMV data and internal records


