Accurate imaging is essential in liver cancer radiation therapy, where precise target definition directly impacts treatment safety and effectiveness. CT remains the standard for simulation and planning, but its limited soft-tissue contrast can hinder tumor delineation, particularly in a dense area of organs at risk and opportunity for tumor motion. MRI overcomes these limitations with superior soft-tissue contrast, improved lesion detection, and the ability to assess and characterize motion of both tumors and adjacent organs at risk. These advantages make MRI a powerful complement or alternative to CT, supporting more accurate gross tumor volume (GTV) definition and greater confidence in treatment planning.
MRI improves simulation and planning in liver cancer radiation therapy
Compared to CT, MRI delivers superior soft-tissue contrast, enabling more accurate delineation of tumors and adjacent organs at risk.¹ This is especially important in the liver, where complex vascular structures and subtle lesion boundaries can complicate GTV definition. MRI’s high-resolution imaging supports more precise and confident contouring—an essential requirement for conformal and stereotactic body radiation therapy (SBRT).
In addition to anatomical clarity, MRI enables motion assessment through techniques such as 4D MRI, which captures respiratory-induced tumor displacement.¹ This information supports individualized motion management strategies—such as gating or breath-hold techniques—and can reduce planning target volume (PTV) margins, improving the therapeutic ratio.
MRI’s superior lesion detection also contributes to more comprehensive radiation treatment planning. In a study of 64 patients with well-differentiated gastroenteropancreatic endocrine tumors, Dromain et al. (2004) found that MRI identified significantly more liver metastases (394) than CT (325) or somatostatin receptor scintigraphy (204), particularly among smaller lesions.² Although diagnostic in nature, the study underscores MRI’s value in providing a complete view of disease burden—critical for accurate gross tumor volume (GTV) delineation. Incorporating MRI into the simulation workflow helps reduce the risk of spatial misses and increases planning confidence, especially in patients with multifocal or ambiguous disease.2 The authors also highlight that healthy liver tissue has a limited tolerance for high-dose radiation, 2 reinforcing MRI’s utility in minimizing additional radiation exposure in simulation versus CT.
MRI supports adaptive planning in liver cancer radiation therapy
MRI also plays a critical role in enabling adaptive radiation therapy for liver cancer. Its superior soft-tissue contrast and accurate lesion mapping allow for meaningful plan adjustments as anatomy changes during treatment cycles.
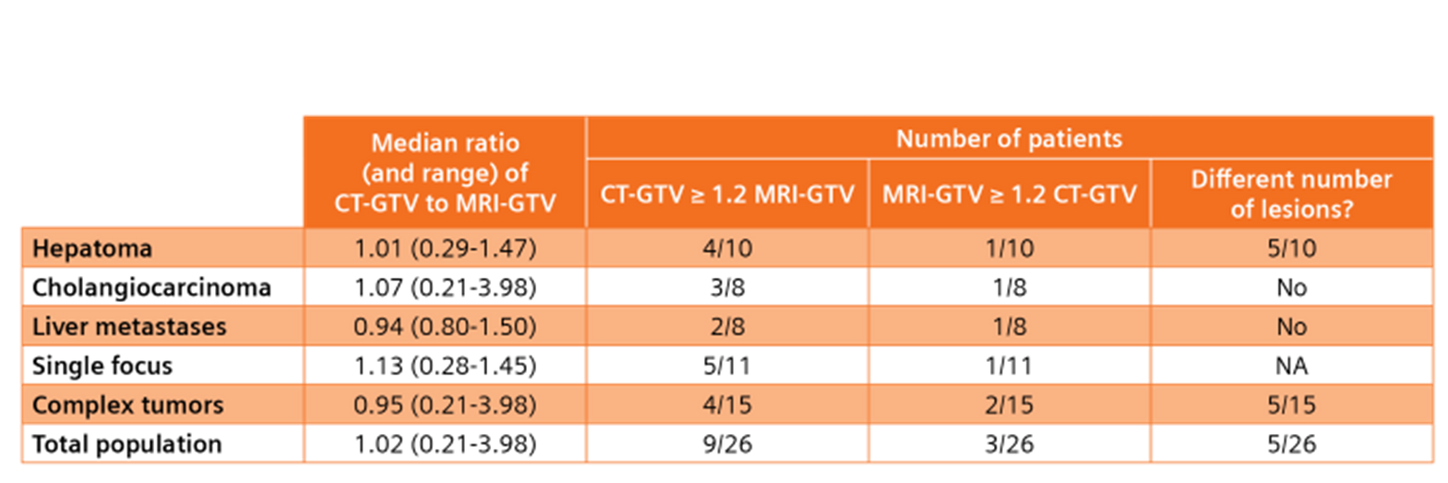
Voroney et al. (2006) examined 26 patients with advanced liver malignancies—including hepatocellular carcinoma (HCC), cholangiocarcinoma, and unresectable metastases—and found that MRI frequently produced GTVs that differed substantially from CT.³ These differences were particularly notable in HCC cases and were often significant enough to impact radiation therapy strategy. Even after applying nonrigid image registration to address respiratory motion, MRI and CT continued to show discordant tumor boundaries. In five HCC patients with large discrepancies, MRI identified additional lesions in three cases.³ The median mismatch in tumor surface area exceeding 5 mm was 26%, and GTV volume concordance ranged from 64% to 81%, depending on tumor type.
These findings underscore MRI’s role in adaptive planning, particularly when standard CT-only workflows fall short in accounting for anatomical variability. As liver tumors and surrounding structures shift during treatment, MRI provides more accurate assessment and informed replanning, whether offline or online.3
Building on these insights, Chen et al. demonstrated that self-gated 4D MRI provides motion-resolved imaging with superior soft-tissue contrast and no ionizing radiation. Compared with 4DCT, 4D MRI more clearly visualized liver and tumor motion, improving internal target volume delineation and supporting precision in adaptive workflows.4
Consider MRI for liver cancer simulation and planning
As liver cancer radiation therapy becomes increasingly precise, image quality during simulation and planning remains critical. While CT may still have its place, MRI provides superior soft-tissue contrast, improved lesion conspicuity, and detailed motion assessment for liver cases. Integrating MRI early in the planning process can reduce the risk of spatial miss, enhance contour accuracy, and enable more adaptive, patient-specific treatment strategies—ultimately supporting safer, more effective radiation therapy.