In head and neck cancers, accurate definition of both the target and nearby organs at risk (OARs) is critical due to the proximity of vital structures and the precision required by conformal treatment approaches. While CT is often the go-to modality for simulation and planning, its limited soft-tissue contrast can compromise contour accuracy—particularly in anatomically complex or infiltrative diseases. MRI provides enhanced soft-tissue differentiation and facilitates more accurate delineation of both tumor boundaries and nearby critical structures. Beyond superior anatomic definition, diffusion-weighted MRI (DWI) provides complementary functional information on tumor cellularity. By quantifying water diffusion through the apparent diffusion coefficient (ADC), DWI helps differentiate viable tumor from necrosis or treatment-related inflammation—an emerging advantage in image-guided radiation therapy planning and assessment. Increasingly, MRI is being integrated into both initial treatment planning and adaptive radiation therapy workflows, where its ability to capture evolving anatomy supports more accurate recontouring and reoptimization throughout the treatment course.
Comparing CT and MRI in simulation and planning for head and neck cancers
Recent studies have begun to clarify the comparative value of CT and MRI for simulation and planning for head and neck cancers. In a direct comparison, Heinke et al. assessed the repeatability of MRI for radiation therapy planning by comparing MRI-to-MRI and CT-to-MRI registration in 50 patients. Using rigid registration of anatomical landmarks, the authors found that MRI-MRI alignment yielded more consistent contours, with parotid and cerebellar volumes appearing smaller on CT than MRI, highlighting MRI’s superior soft-tissue contrast and reliability for accurate structure delineation. Because the repeat scans were acquired under identical setup conditions on the same day, these findings demonstrate MRI’s stability for planning.1
Beyond improved anatomic visualization, DWI can also provide biologic insight. DWI offers tissue characterization that complements CT’s density-based data. Tumor regions demonstrating restricted diffusion and low ADC values correlate with higher cellularity, while post-treatment necrosis and edema exhibit elevated ADC. Incorporating DWI alongside T1- and T2-weighted sequences may therefore refine GTV definition, particularly in areas of heterogeneous signal or uncertain viability.2
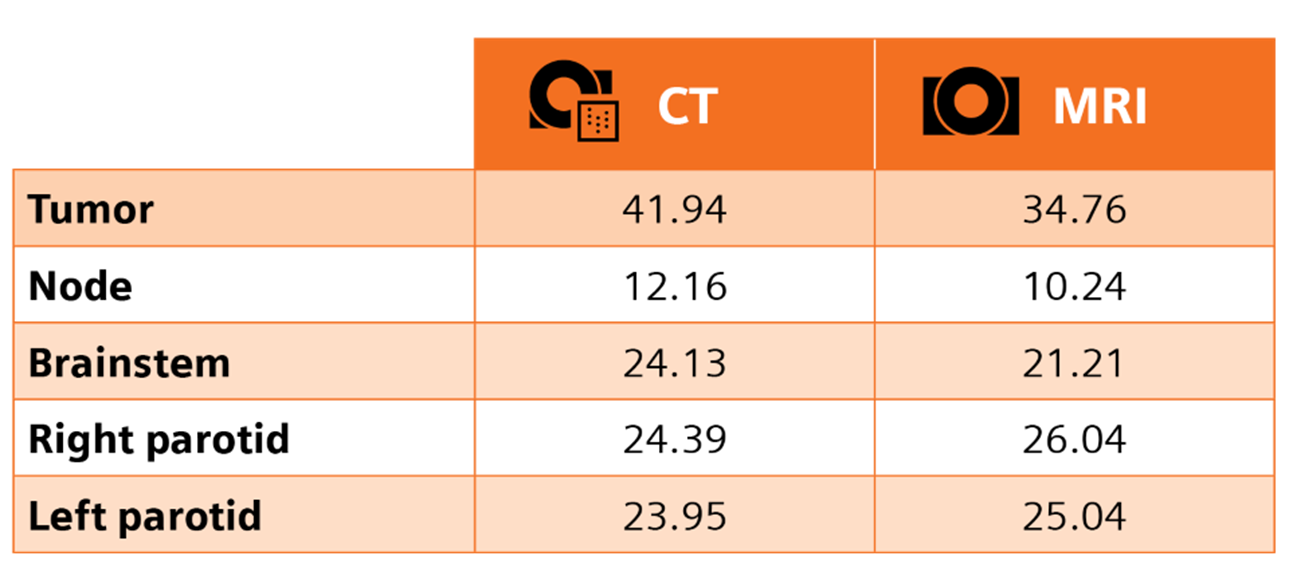
Lekshmi et al. reviewed the CT and MRI scans of contoured primary tumors, lymph nodes and OARs of 54 patients. The MRI scans showed larger parotid gland volumes than that on the CT scans while MRI-based volumes for the primary tumor, nodes, and brainstem were all smaller than CT. By more clearly defining healthy tissue versus tumors, radiation oncologists could reduce the chances of toxicity in healthy tissue and provide better tumor control.3
MRI supports adaptive planning in liver cancer radiation therapy
MRI also plays a critical role in enabling adaptive radiation therapy for liver cancer. Its superior soft-tissue contrast and accurate lesion mapping allow for meaningful plan adjustments as anatomy changes during treatment cycles.
Voroney et al. (2006) examined 26 patients with advanced liver malignancies—including hepatocellular carcinoma (HCC), cholangiocarcinoma, and unresectable metastases—and found that MRI frequently produced GTVs that differed substantially from CT.³ These differences were particularly notable in HCC cases and were often significant enough to impact radiation therapy strategy. Even after applying nonrigid image registration to address respiratory motion, MRI and CT continued to show discordant tumor boundaries. In five HCC patients with large discrepancies, MRI identified additional lesions in three cases.³ The median mismatch in tumor surface area exceeding 5 mm was 26%, and GTV volume concordance ranged from 64% to 81%, depending on tumor type.
Table 1. Mean structure volumes (cc) on CT vs. MRI in head and neck cancer (N=54).
In another study of 35 nasopharyngeal cancer patients, Gong et al. found that MRI offered clearer visualization of soft-tissue structures—such as the parotid glands, brainstem, and optic nerve—compared to CT. MRI-based volumes of the parotid glands were 7–8% larger, leading to a statistically significant 4–5 Gy increase in mean dose. The brainstem appeared 9.33% smaller on MRI, resulting in a 4.32 Gy reduction to its most exposed 0.1 cm³. While CT remained reliable for evaluating bony and well-defined structures, MRI was favored for delineating soft-tissues for certain cancers and by creating more accurate images, supporting improved dose and treatment planning.4
Accurate nodal staging plays a critical role in managing head and neck squamous cell carcinoma, especially in patients who don’t have any clinically palpable cervical lymphadenopathy (N0 neck). While palpation is still a routine part of the clinical exam, it misses cervical lymph node metastases in about 20–28% of cases. Cross-sectional imaging with CT, especially contrast-enhanced CT (CECT), improves detection but reported miss rates range from approximately 8% to 28%, influenced by size criteria, imaging protocols, and reader experience. MRI tends to be more consistent, showing an average staging error of around 16%. This suggests that MRI may offer more reliable detection than CT in certain situations—especially in anatomically complex regions or when trying to pick up on smaller nodal disease.5
Tracking anatomical change: MRI’s role in adaptive therapy
While MRI improves accuracy at simulation, its value extends well beyond initial planning. In head and neck cancers, anatomical structures can change significantly over the course of treatment, affecting both target volumes and OARs. These dynamic shifts make a strong case for adaptive radiation therapy (ART)—and MRI is uniquely positioned to guide it.
Morgan et al. conducted a prospective study using weekly MRI to evaluate volumetric changes in primary tumors and involved lymph nodes during chemoradiation for head and neck squamous cell carcinoma. They observed significant and progressive reductions in tumor (16%, 48%, 66%) and nodal volumes over weeks 2, 4, and 7, with substantial inter-patient variability and no reliable predictors of individual response. These findings highlight the dynamic nature of tumor anatomy during treatment and support the integration of MRI in adaptive radiation therapy. By improving dose delivery and minimizing unnecessary exposure to surrounding OARs, MRI enhances the safety and efficacy of treatment delivery.6
Emerging evidence suggests that serial DWI and ADC mapping could enhance adaptive workflows by serving as quantitative biomarkers of treatment response. In prospective studies, early increases in ADC during chemoradiation have correlated with favorable local control, while persistently low or declining ADC may indicate resistant disease. These findings underscore DWI’s potential to inform bioadaptive replanning, integrating functional response metrics into anatomical adaptation and moving toward more personalized, data-driven radiation therapy.2
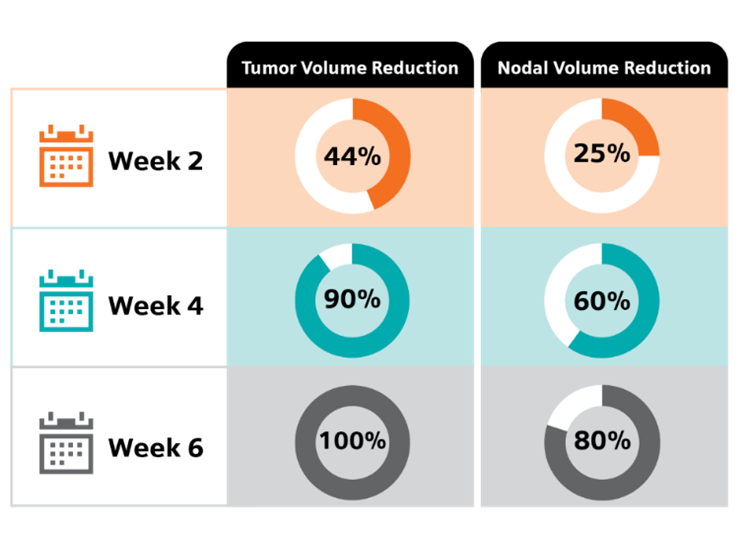
To assess the clinical impact and timing of MRI in ART, a feasibility study at MD Anderson Cancer Center followed five patients with locally advanced oropharyngeal cancer. Biweekly MRI scans revealed marked tumor shrinkage—44% at week 2, 90% at week 4, and complete resolution by week 6. Nodal volumes also declined significantly, with reductions of 25%, 60%, and 80% at the same time points. These rapid and dynamic anatomical changes impacted dose distribution, prompting adaptive replanning that reduced the mean parotid dose by 3.3 Gy. While modeling showed only a modest 1% reduction in xerostomia risk, the projected risk of dysphagia dropped by 11% at six months, underscoring the clinical value of MRI in ART in sparing healthy tissue and tailoring treatments to evolving anatomy.7
Tumor and Nodal Volume Reduction Over Time7
Bringing the clinical value of MRI to the forefront
As radiation oncology continues to evolve toward more precise and personalized treatment, MRI’s clinical advantages in head and neck cancers are increasingly clear. Superior soft-tissue contrast enhances the accuracy of tumor and OAR delineation—especially in anatomically complex regions where millimeters matter. Studies demonstrate that MRI supports more reliable staging, more precise volume definition, and more responsive adaptive planning than CT. By enabling better sparing of critical structures such as salivary glands, brainstem, and pharyngeal constrictors, MRI helps reduce toxicity and preserve essential functions like speech and swallowing. As MRI advances further in radiation therapy, incorporating functional sequences such as DWI may strengthen biologic understanding of tumor behavior, linking imaging phenotype with dose adaptation, response prediction, and long-term outcomes.