T-trendz Issue 2 / 2021
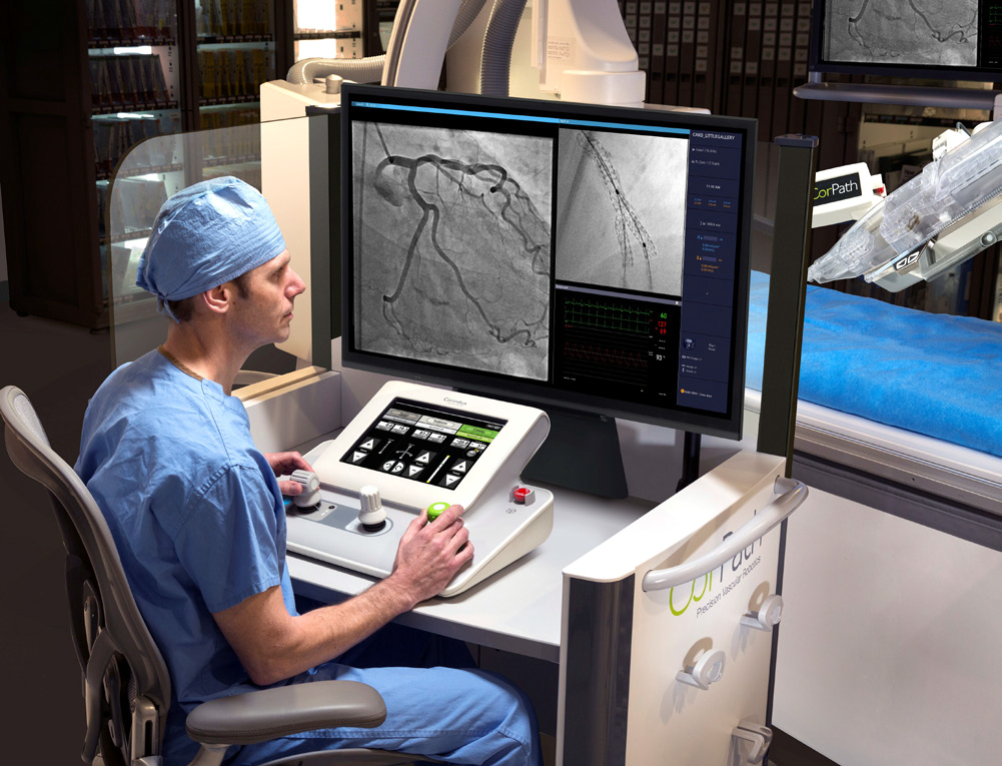
Explore the benefits of Robotics in Vascular Interventions with Corindus
#This study was performed at a single center and there can be no guarantee that other customers will achieve the same results.
1Mahmud E, et al. Demonstration of the Safety and Feasibility of Robotically Assisted Percutaneous Coronary Intervention in Complex Coronary Lesions Results of the CORA-PCI Study (Complex Robotically Assisted Percutaneous Coronary Intervention). JACC Cardiovascular Interventions. 2017 Jul 11;10(13):1320-1327; DOI: 10.1016/j.jcin.2017.03.050.
2Patel T, et al. Comparison of Robotic Percutaneous Coronary Intervention With Traditional Percutaneous Coronary Intervention. Circ Cardiovasc Interv. 2020; 13:e008888.”
3Campbell PT, et al. The impact of precise robotic lesion length measurement on stent length selection: Ramifications for stent savings. Cardiovasc Revasc Med. 2015 Sep;16(6):348‐50. doi:10.1016/j. carrev.2015.06.005. Epub 2015 Jul 9.
4Weisz G, et al. Safety and Feasibility of Robotic Percutaneous Coronary Intervention ‐ The Multi‐Center Percutaneous Robotically‐Enhanced Coronary Intervention Study (PRECISE). Journal of the American College of Cardiology. 2013 Apr 16;61(15): 1596‐600. doi: 10.1016/j.jacc.2012.12.045.
5Mahmud et al. “TCT-442 Robotic Peripheral Vascular Intervention with Drug Coated Balloons is Feasible and Reduces Operator Radiation Exposure: Results of the Robotic-
Assisted Peripheral Intervention for Peripheral Artery Disease (RAPID) Study II.” JACC, Vol 72 No. 13: 2018.
6Data gathered using CorPath 200.
7Weisz G, et al. Safety and Feasibility of Robotic Percutaneous Coronary Intervention - The Multi-Center Percutaneous Robotically-Enhanced Coronary Intervention Study (PRECISE).
Journal of the American College of Cardiology.2013 Apr 16;61(15): 1596-600. doi: 10.1016/j.jacc.2012.12.045