- Home
- About Siemens Healthineers India
- T-trendz
- T-trendz Issue 1 / 2021
T-trendz Issue 1 / 2021
Achieve consistent high-quality results in neuro-interventions with syngo DynaCT and syngo Dyna4D
syngo DynaCT – (Cone Beam CT)

syngo DynaCT is an advanced imaging technique which creates a 3D data set from the series of images acquired from various projections of rotational angiography around the patient. Conventionally it delivers 3D images with high-contrast details of bones, clips, coils, and vessels. syngo DynaCT imaging is the result of the advanced development of 3D high-contrast imaging but processed to visualize low-contrast soft tissue information. It produces CT like quality in the angio suite with workstation-based software applications connected to main cath lab system equipped with large size flat detectors.
syngo DynaCT increases the level of confidence to physicians by providing cone beam CT when it is used as an adjunct to DSA or fluoroscopy. In general, the assessment by syngo DynaCT adds value to existing technologies like dynamic flat detector in the interventional suite by providing soft tissue information. This enables the treating physician to plan more effective interventions, and results in reduction of treatment-related complications.

What is ‘soft tissue information’?
With syngo DynaCT, it is possible to achieve a higher level of tissue differentiation in the reconstructed images, enabling the visualization of objects with lower Hounsfield Units (HU) values. You can visualize objects of 5 HU and 10 mm in size or 10 HU and 5 mm in size(as measured using a 16 cm CATPHAN phantom). Thus, enabling visualization of lower contrast objects such as larger hemorrhages and tumors.
Watch video: Easy steps to 3D/DynaCT results
How does syngo DynaCT work?
syngo DynaCT utilizes images acquired from a special rotational angiography run to reconstruct cross-sectional soft tissue information. Image acquisition is achieved in 5-20 seconds, depending on the protocols used. During that time the C-arm covers an angle of at least 200° and acquires the needed projection images for reconstruction. The Rotation speed of C-arm related to protocol may vary from 10 to 40 degrees per second.
Visualization modes
The result of a 3D rotational angiography is a stack of images that can be visualized either through MPR (Multiplanar Reformation/ Reconstruction), thick MPR, MIP (Maximum Intensity Projection) or through VRT (Volume Rendering Technique).
What is the spatial resolution of syngo DynaCT and what slice thickness can be reconstructed? The slice thickness that can be reconstructed depends on the volume of interest and the voxel size. The standard slice thickness is about 0.5 mm and there are additional reconstruction options that allow creation of lower slice thickness. The spatial resolution achieved with syngo DynaCT is about 0.22 mm.
Advantages
The primary advantages over conventional CT are ability to focus in detail on a small area surrounding tissue, lower operational cost and smaller footprint, saving precious time during interventions. In addition, three-dimensional rotational angiography combines the accuracy of direct angiography with the benefits of computer animation and supplements conventional 2D fluoroscopy to provide real-time representation of structures (syngo 3D Road-mapping). Although this fusion is possible with other modality images as well, syngo DynaCT gives the advantage of reducing the dose which usually is not the case during registration of images from other modalities.
Watch video: DynaCT acquisition movement
syngo Dyna4D
In interventional radiology and neuroradiology, getting the best possible diagnosis is central to a therapy’s success – before, during, and on completion of the intervention. This allows the physician to plan and perform the treatment precisely. In contrast to current 3D image acquisition methods, syngo Dyna4D uses a slightly modified protocol, which makes it possible to combine spatial and temporal resolution (“3D+t”).
Case Study 1: Treating Right ICA blister aneurysm with flow diverting stent using single plane system with syngo DynaCT
Dr. Barath Sitaram
Geetanjali Hospital, Udaipur, India

Assistant Professor / Consultant Neuro & Vascular Interventional Radiologist
Image 1.3: DynaCT stent recon with diluted contrast injection showing flow divertor wall apposition was confirmed with DynaCT run
Image 1.1: 3D High contrast image showing right ICA blister aneurysm
Image 1.2: Post stenting DynaCT of CMPR cross section of stent
Image 1.3: DynaCT stent recon with diluted contrast injection showing flow divertor wall apposition was confirmed with DynaCT run
Image 1.1: 3D High contrast image showing right ICA blister aneurysm
Image 1.2: Post stenting DynaCT of CMPR cross section of stent
Image 1.3: DynaCT stent recon with diluted contrast injection showing flow divertor wall apposition was confirmed with DynaCT run
35-year-old female was admitted with severe headache. CT scan showed grade 2 sub-arachnoid hemorrhage. DSA Angio was performed and showed right ICA blister aneurysm which was confirmed with 3D reconstruction (5 sec DSA, 3D run).
Patient was treated with flow divertor implantation under GA. Later Flow divertor wall apposition was confirmed with 20 sec DynaCT run.
For any further discussions about the case please write to barath.sitaram@yahoo.com
Case Study 2: Treating ethmoidal dural AVF using biplane system with syngo DynaCT
Dr. Manish Shrivastava
KDA Hospital, Mumbai, India

Head, Interventional Neuroradiology
46-year-old Indian male presented with headache, vomiting and seizure. Cross sectional imaging with angiogram was advised for both followed by Digital Subtraction Angiography and Embolisation with non-adhesive liquid embolic agent (Squid) of anterior cranial fossa (ethmoidal) dAVF via transarterial access.

Image 2.3: DynaCT confirms good deposition of squid, done via facial artery
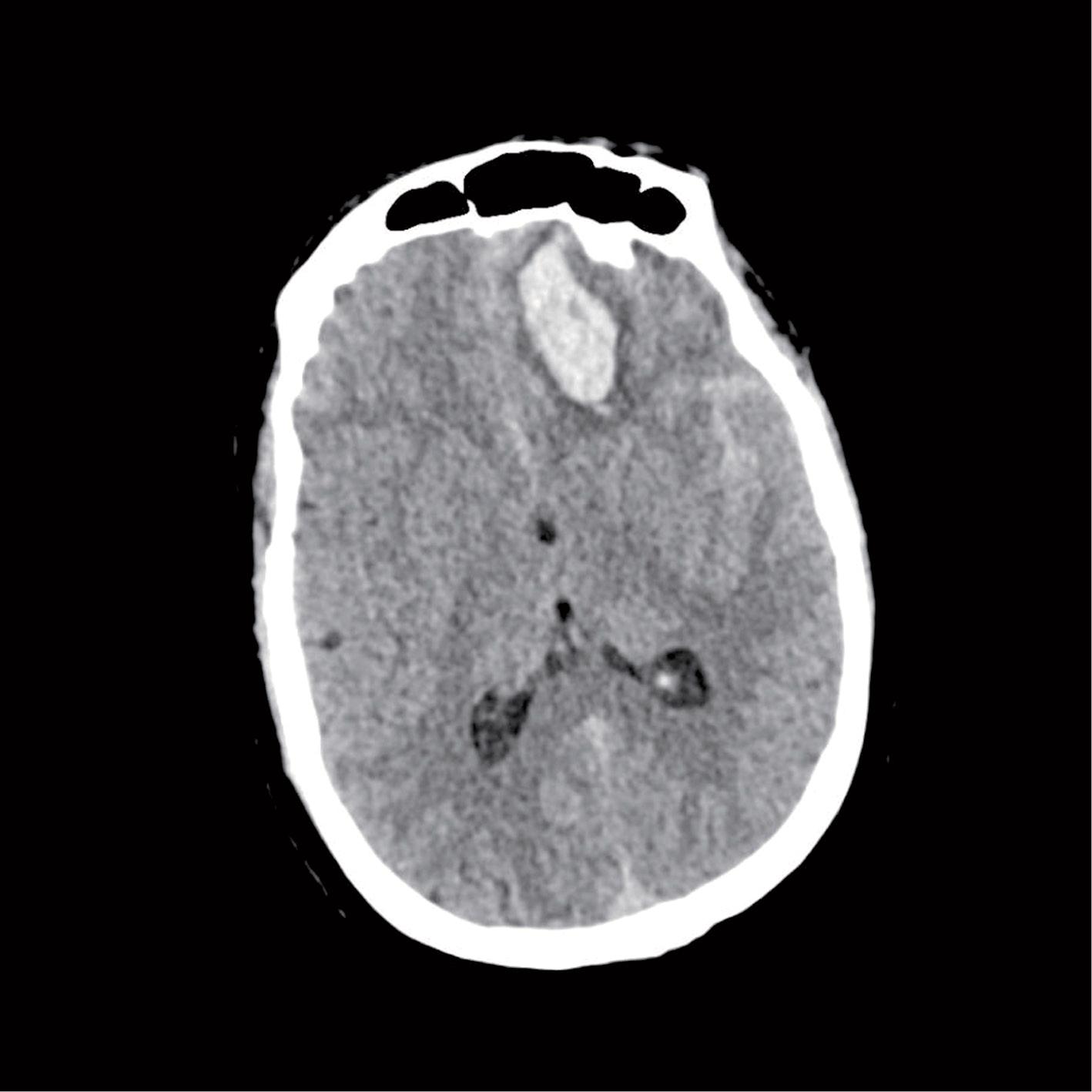
Image 2.1: Left frontal hematoma with SAH

Image 2.2: ECA angiogram shows feeders from facial artery branch angular artery

Image 2.3: DynaCT confirms good deposition of squid, done via facial artery

Image 2.1: Left frontal hematoma with SAH

Image 2.2: ECA angiogram shows feeders from facial artery branch angular artery

Image 2.3: DynaCT confirms good deposition of squid, done via facial artery
On cross sectional imaging it showed unilateral frontal lobe hemorrhage with abnormal feeding vessels adjacent to it. Digital Subtraction Angiography confirmed Ethmoidal Dural AVF. Embolisation with non-adhesive liquid embolic agent of anterior cranial fossa (ethmoidal) dAVF via transarterial (angular artery) access was performed. After embolization, cerebral angiography showed no arteriovenous shunt, compatible with radiographic cure of the fistula. Dyna CT performed showed squid cast along AVF and venous ectasia, confirming correct distribution of embolic agent. The patient recovered fully and were discharged home three weeks after the procedure. They returned for their one-month follow-up appointment without complaint and neurologically intact.
Ethmoidal dural arteriovenous fistulas (AVFs) are rare intracranial lesions associated with a high risk of intracranial hemorrhage. It comprises about 5% of all intracranial dAVFS. They are unique in two ways - they always have retrograde leptomeningeal venous drainage and they are frequently supplied by a fine tuft of feeders arising from anterior ethmoidal branches of the ophthalmic arteries. Dural arteriovenous fistulas (dAVFs) of the anterior cranial fossa have traditionally been treated by open surgical disconnection. Safe navigation through the ophthalmic artery or fragile cortical veins has historically provided a barrier to effective endovascular occlusion of these lesions. Using current microcatheter technology, embolic materials and DynaCT images safe positioning within the distal ophthalmic artery, beyond the origin of the central retinal artery, is achievable.
For any further discussions about the case please write to docmanishs@yahoo.co.in
Case Study 3: Treating deep seated compact nidus arteriovenous malformation using biplane with 4D rotational angiogram
Dr. Mathew Cherian
KMCH, Coimbatore,, India

Chief of Radiology, Consultant Vascular and
Interventional Radiology

Image 3.1: Axial non-contrast CT of the brain at the level of basal ganglia shows a large acute intracerebral bleed in the right basal ganglia region
with mass effect and midline shift
A 39-year-old female with no co-morbidities, presented to the emergency with history of sudden onset severe headache, vomiting and left sided weakness. Neuroimaging revealed acute right basal ganglionic hematoma with mass effect and mild midline shift. A cerebral angiogram revealed a deep-seated compact nidus arteriovenous malformation measuring about 4.2 x 4 cm in diameter in the right basal ganglia region. Deep seated AVMs pose significant challenges to treatment and cure. Risk of stroke and periprocedural bleeding are higher compared to a superficially located AVM. Hence, a 4D rotational angiogram was performed to understand the angio-architecture and plan treatment. The temporal resolution of this module allows us to clearly separate the feeders, the nidus and the draining veins in three dimensions, making it the most informative study for the angio-architecture of the AVM. High risk vascular features like feeding artery/ intranidal aneurysms, deep venous drainage and draining vein stenosis can be picked up with great ease.
In our case, the AVM was fed by a dominant large perforator from the right middle cerebral artery M1 segment and multiple small en-passage feeders from the M2 and M3 segments. Two pre-nidal irregular feeding artery aneurysms were noted. The proximal aneurysm measured 2.85 x 2.3 x 2.5 mm (FxHxN) and the distal aneurysm measured 3.8x3.5x2.8 mm (FxHxN). Two intranidal aneurysms were also seen measuring 5.3x7.5x3.8 mm and 3.2x4x3.2 mm (FxHxN), one of their domes facing the haematoma. Deep venous drainage was noted through ectatic veins into the right cavernous sinus and the straight sinus via the right internal cerebral vein. Cortical venous reflux is seen into the middle cerebral vein. There was no evidence of draining vein stenosis.
Video: 4D rotational angiogram of the right ICA in various phases of the AVM filling show feeding artery (arrow) and intranidal (thick arrow) aneurysms and the deep venous drainage
After a multi-disciplinary discussion, we decided to stage the treatment and address the high risk anatomical features in the first stage by targeted embolization. The 4D rotational angiogram provided a detailed picture of the feeding artery (hypertrophied M1 perforator) and intranidal aneurysms and thus we could find working angles which best depicted them. Targeted embolization of the feeding artery carrying the aneurysms along with the intranidal aneurysms was performed with a Sonic (Balt) detachable tip (3 cm) microcatheter using Menox 18 (Meril). Post embolization, the patient underwent rehabilitation and was discharged with partial improvement of left sided paresis.
The patient was planned for stereotactic radiosurgery after recovery considering the deep location and angio-architecture.
For any further discussions about the case please write to Dr.mathewcherian@gmail.com
Technological innovations spur new clinical applications. This gives healthcare providers an edge in detection and treatment of diseases at an early stage with minimally invasive techniques. This in turn will help in expanding precision medicine, transforming care delivery and improving patient experience. These are the primary objectives with which we developed T-trendz. We would like to know how we can make this initiative more valuable for your practice and the wellbeing of patients.
© Siemens Healthcare GmbH, 2021
All trademarks are properties of Siemens Healthcare GmbH or associate companies. Third-party trademarks are properties of their respective companies. The content of this publication does not necessarily reflect the opinion of the publisher. The information is provided by Registered Medical Practitioners or Hospital Staff only and is not an advice and should not be treated as such. Siemens Healthcare GmbH or associate companies make no representations or warranties of any kind, express or implied, about the completeness, accuracy, reliability, suitability or availability with respect to the website or the information, products, services, or related graphics contained on the website for any purpose. Any reliance you place on such information is therefore strictly at your own risk. Reproduction of articles in whole or in part requires the permission of Siemens Healthcare GmbH or associate companies and the respective Registered Medical Practitioners or Hospital Staff and in this due course, Siemens takes no responsibility for the intent of the article being preserved post such alterations or modifications.. This also applies to storage in electronic databases and on the internet.
It is intended for use by registered medical practitioners or hospital staff only. The medical information in this newsletter is for individuals of the healthcare community and not for general public. The information and reference materials contained here are intended solely for the general information of the reader and is neither intended to dictate what constitutes reasonable, appropriate or best care for any given health issue, nor is it intended to be used as a substitute for the independent judgment of a physician or a surgeon for any given health issue.