
Cios Spin mobile C-arm and NuVasive Pulse platform
Navigation and excellent intraoperative imaging in spine surgery offer the potential for more surgeons to adopt MIS techniques, while improving implant accuracy and minimizing radiation. However, the complexity of existing solutions and the workflow disruptions they cause have impeded adoption of navigation in the majority of spine surgeries.
Siemens Healthineers and NuVasive formed the Spine Precision Partnership to create a holistic solution for spine surgery.
Cios Spin and the Pulse platform now integrate seamlessly, delivering 2D and 3D imaging, navigation technologies, image quality improvement for low dose images, and a wide range of dedicated spine surgery technologies — all available on one easy-to-use platform.

Cios Spin performs an isocentric 3D scan in 30 seconds
Cios Spin: Mobile 3D imaging for intraoperative quality assurance
Retina 3D: Precise 3D visualization with intraoperative cone beam CT
Retina 3D lets you see fine anatomical structures, implants, screws, and devices. The 3D technology of Cios Spin may also help to avoid revision surgery.
Easy 3D: 3D made quick and easy
With step-by-step guidance, scan times of only 30 seconds, and easy-to-use 3D visualization functions, Cios Spin improves 3D workflow efficiency.
Screw Scout2: Accelerated intraoperative screw assessment
Screw Scout automatically localizes screws in the 3D volume and presents the detected screws in a centered view in three planes. This allows for immediate and accelerated screw assessment.
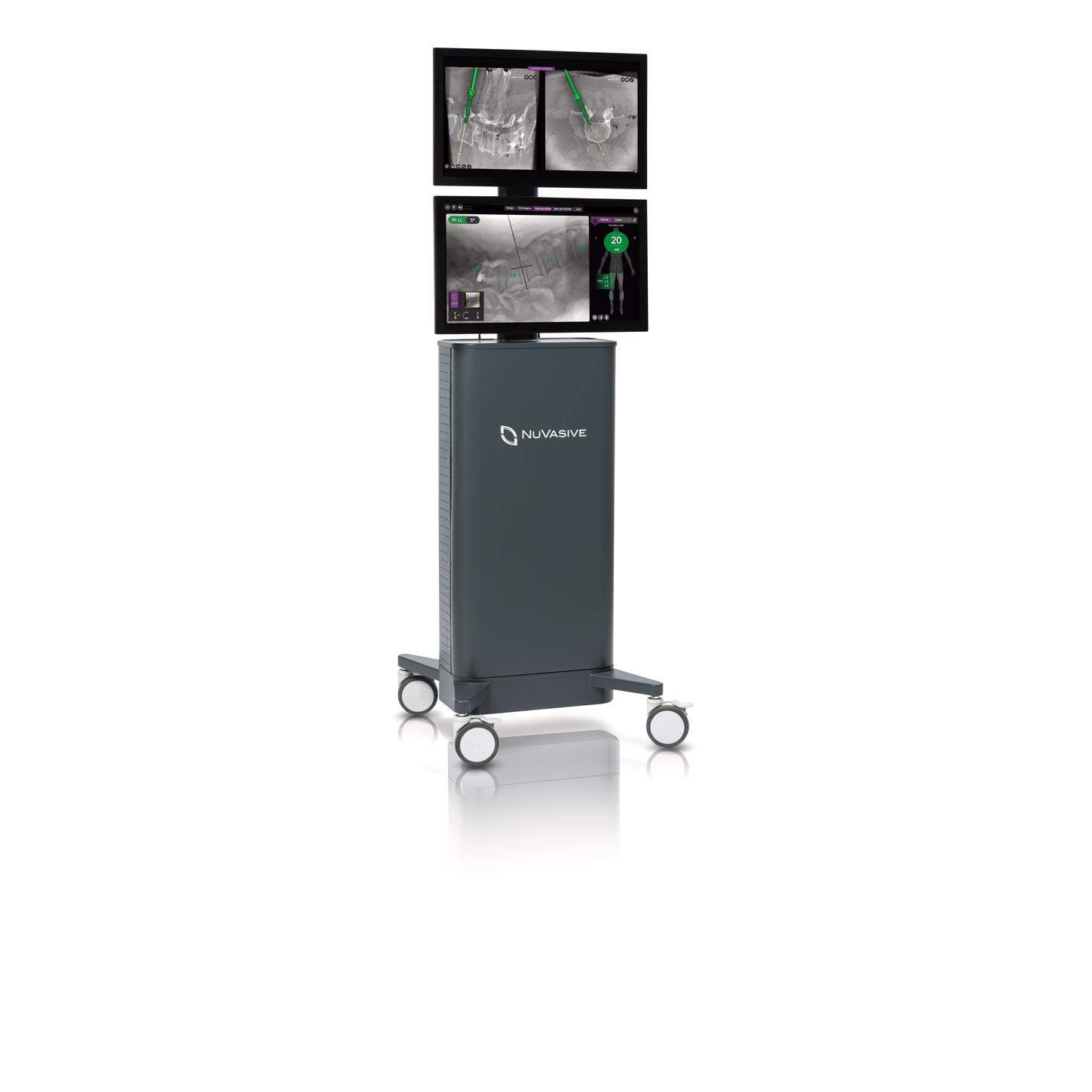
NuVasive Pulse platform
Pulse: An integrated technology platform to enable better spine surgery
Precision through 3D imaging and navigation
Pulse introduces a procedurally integrated navigation technology that features workflow technologies to improve ease-of-use and surgical efficiency in the OR.
Smart imaging with radiation reduction
The Lessray technology was designed to increase OR efficiency through a streamlined imaging workflow, and to reduce radiation exposure for everyone in the room.4
Multiple technologies in one platform
Besides navigation and Lessray, Pulse integrates multiple enabling technologies to improve workflow in the OR, including neuromonitoring, Integrated Global Alignment, and Bendini.
“Using both systems connected to each other, we have so many features dedicated to spine surgery, which provides more effective and meaningful delivery of information in the OR with a small equipment footprint within the sterile field.”
Customer testimonial
References
Beisemann N, Gierse J, Mandelka E, Vetter S, et al. Comparison of three imaging and navigation systems regarding accuracy of pedicle screw placement in a sawbone model. Sci Rep 12, 12344 (2022).
Foster N, Shaffrey C, Buchholz A, et al. Image Quality and Dose Comparison of 3 Mobile Intraoperative Three-Dimensional Imaging Systems in Spine Surgery. World Neurosurgery, Volume 160, 2022, Pages e142-e151. ISSN 1878-8750.
Eastlack R, Mundis G, Uribe J, Mimran R, Kretzer R, Hassanzadeh H, et al. An Evaluation of the Accuracy, Efficiency, and Safety of Pulse® Navigation Percutaneous Pedicle Screw Placement: A Randomized, Cadaveric Study. NuVasive, Inc. November 2021. 9513871 B.





