Siemens Healthineers History of ICE Innovation

2000
In 2000, the first ACUSON AcuNav 10 French catheter was introduced to the market. This was the first phased array intracardiac echocardiography (ICE) catheter. It enabled improved visualization for heart applications in electrophysiology and interventional cardiology.

2004
In 2004, ACUSON introduced the smaller 8 French catheter to better meet the pediatric interventionalist's needs.

2006
In 2006, Siemens Healthineers partnered with Biosense Webster, A Johnson & Johnson Company (leading manufacturer of electrophysiology mapping equipment and therapeutic & diagnostic catheters) to further advance the ACUSON AcuNav catheter into the area of imaging during EP procedures.

2007
2007 brought the introduction of the SoundStar® catheter. This technology integrated the ACUSON AcuNav catheter technology with sensor technology to enable EP mapping technology, which accurately places data sets from the 2D ICE image into the X Y Z planes, creating three-dimensional images of the heart chambers. The CartoSound® technology (a Siemens Healthineers/BWI collaboration)1 is a differentiating technology in electro-anatomical mapping.

2012
In 2012, Siemens Healthineers introduced the first real-time volume ICE catheter, the ACUSON AcuNav V catheter. The catheter was a 90° x 24° imaging catheter created to address the imaging needs of the structural heart environment where cross-sectional views of the valve orifices are required to visually guide valve repair and replacement procedures.
2017
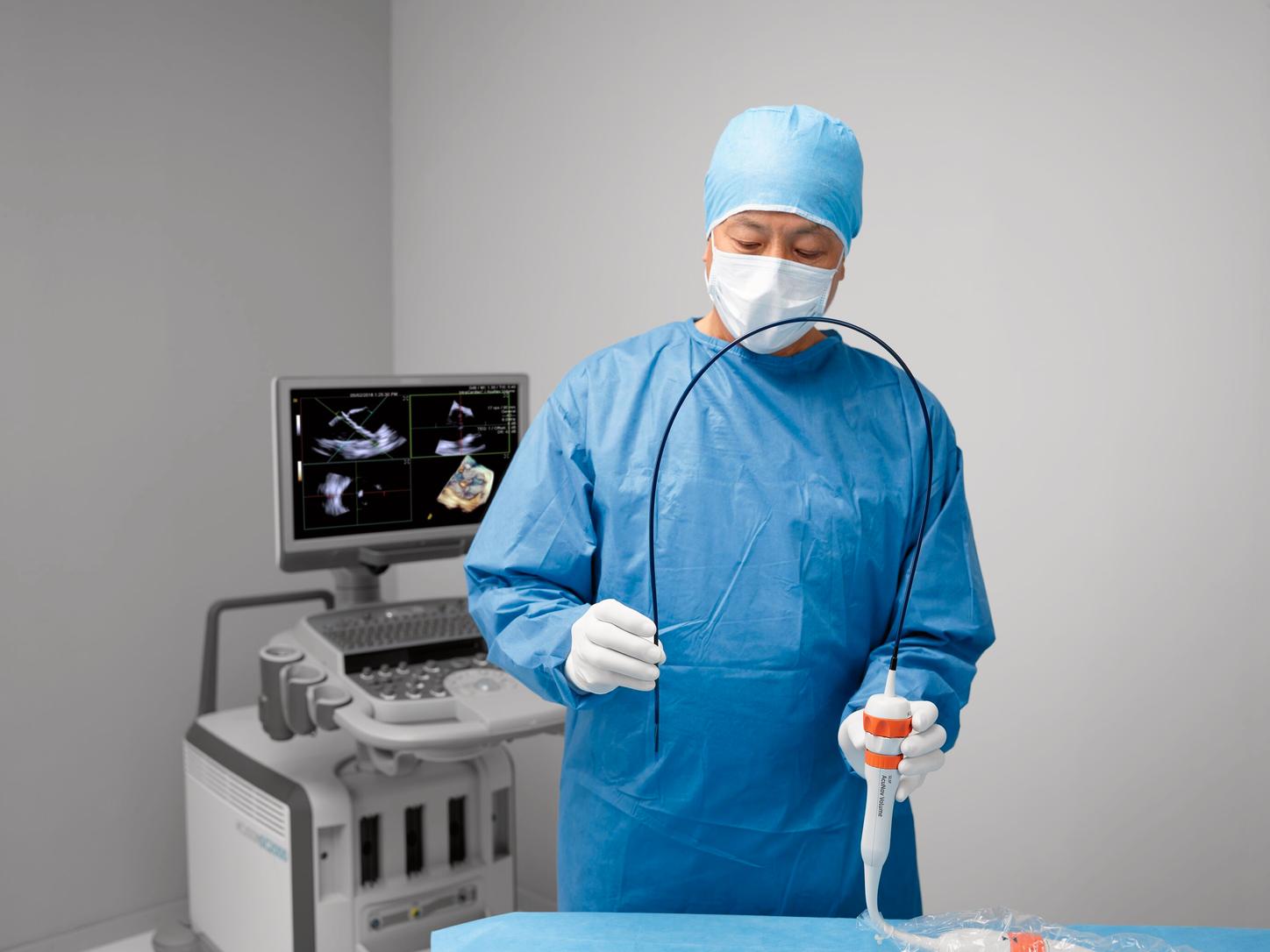
In 2017, Siemens Healthineers received FDA clearance for the ACUSON AcuNav Volume ICE catheter, the next generation of volume (4D) ICE catheter imaging. The catheter brings a broader elevation plane than the ACUSON AcuNav V catheter and provides PW, CW, and color Doppler capabilities in both 2D and 4D imaging modes.
2022
In 2022, working closely with customers, Siemens Healthineers continued the tradition of innovation and released an updated version of the AcuNav Volume 4D ICE catheter – with an improved imaging approach.
ICE Clinical Case Studies
Novel Approach to LAAO Procedures
This case of left atrial appendage occlusion (LAAO) with a Watchman device (Boston Scientific) demonstrates the advantages of using the ACUSON AcuNav Volume ICE Catheter over transesophageal echocardiography (TEE) and 2D ICE.
MitraClip™ Deployment with ACUSON AcuNav Volume ICE Catheter
A 38 year old female with significant osteogenesis imperfecta, severe scoliosis and severe myxomatous mitral valve insufficiency from bileaflet prolapse was referred for MitraClip. She had an additional history of COPD with CO2 retention and obstructive sleep apnea. She had been experiencing dyspnea, consistent with NYHA III symptoms.
An adult TEE transducer could not be advanced due to the severe scoliosis and pediatric TEE imaging was suboptimal for use in the procedure. TTE, fluoroscopy and volume ICE were used to successfully deploy a MitraClip.