- Home
- Medical Imaging
- Molecular Imaging and Nuclear Medicine Systems
- Radiopharma
- European PET Tracers | Summaries of Product Characteristics
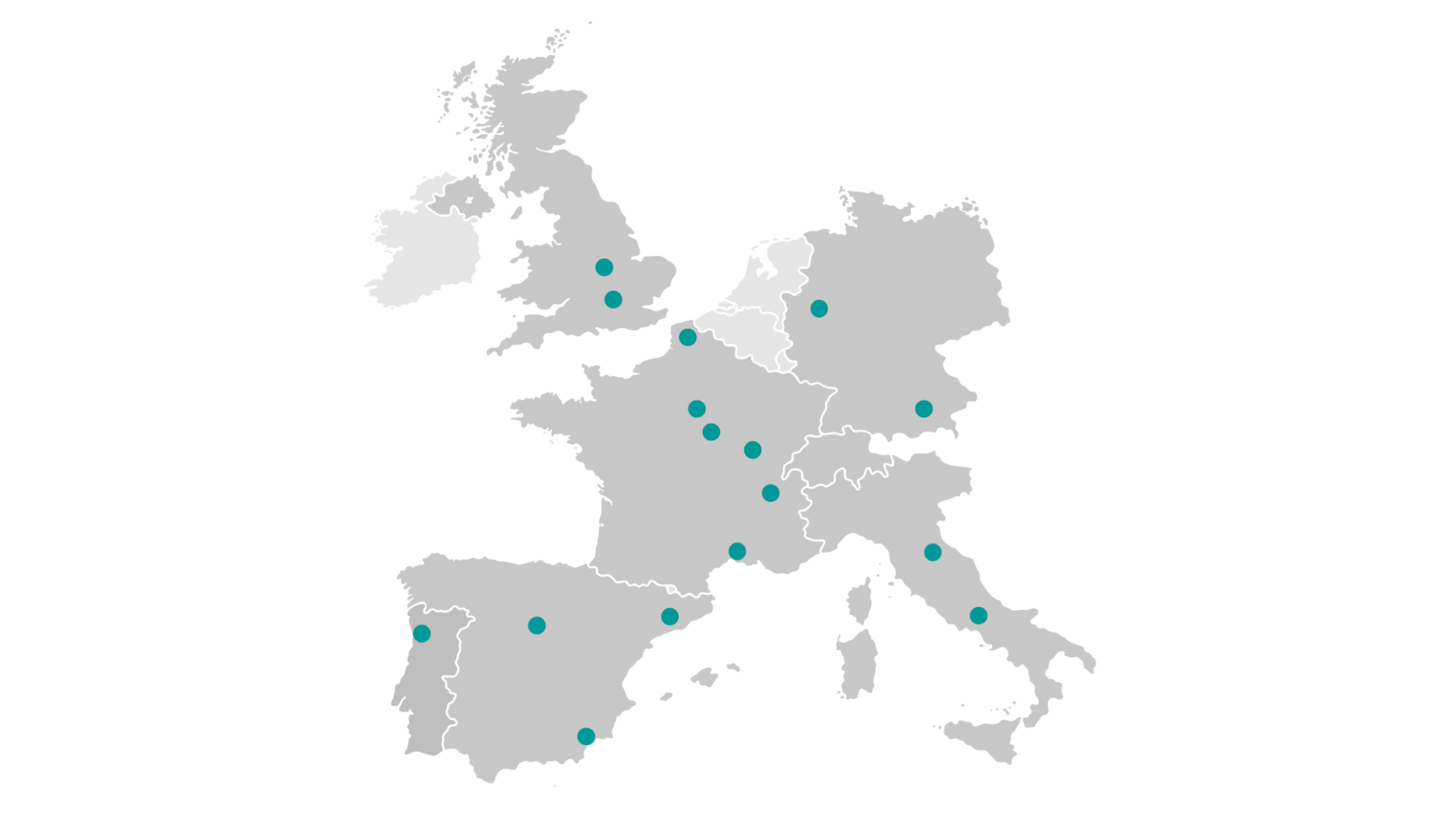
European PET tracers
Summaries of product characteristics Broadest portfolio of PET tracers from a single supplier
Product portfolio
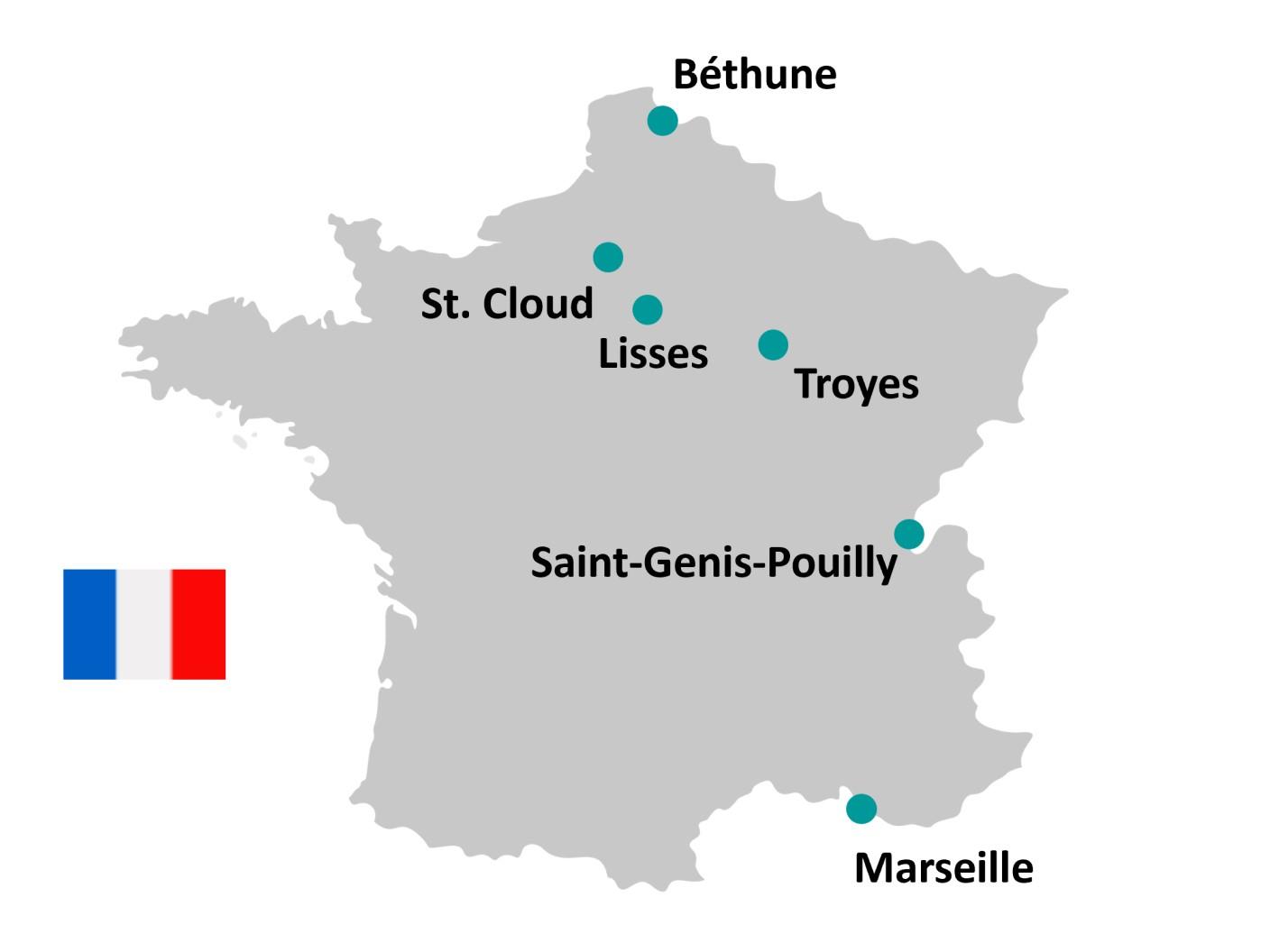
Available PET radiopharmaceuticals
Gluscan
Fluorochol
Additional PET radiopharmaceuticals are available, please contact your local pharmacy.
Available PET radiopharmaceuticals
Additional PET radiopharmaceuticals are available, please contact your local pharmacy.
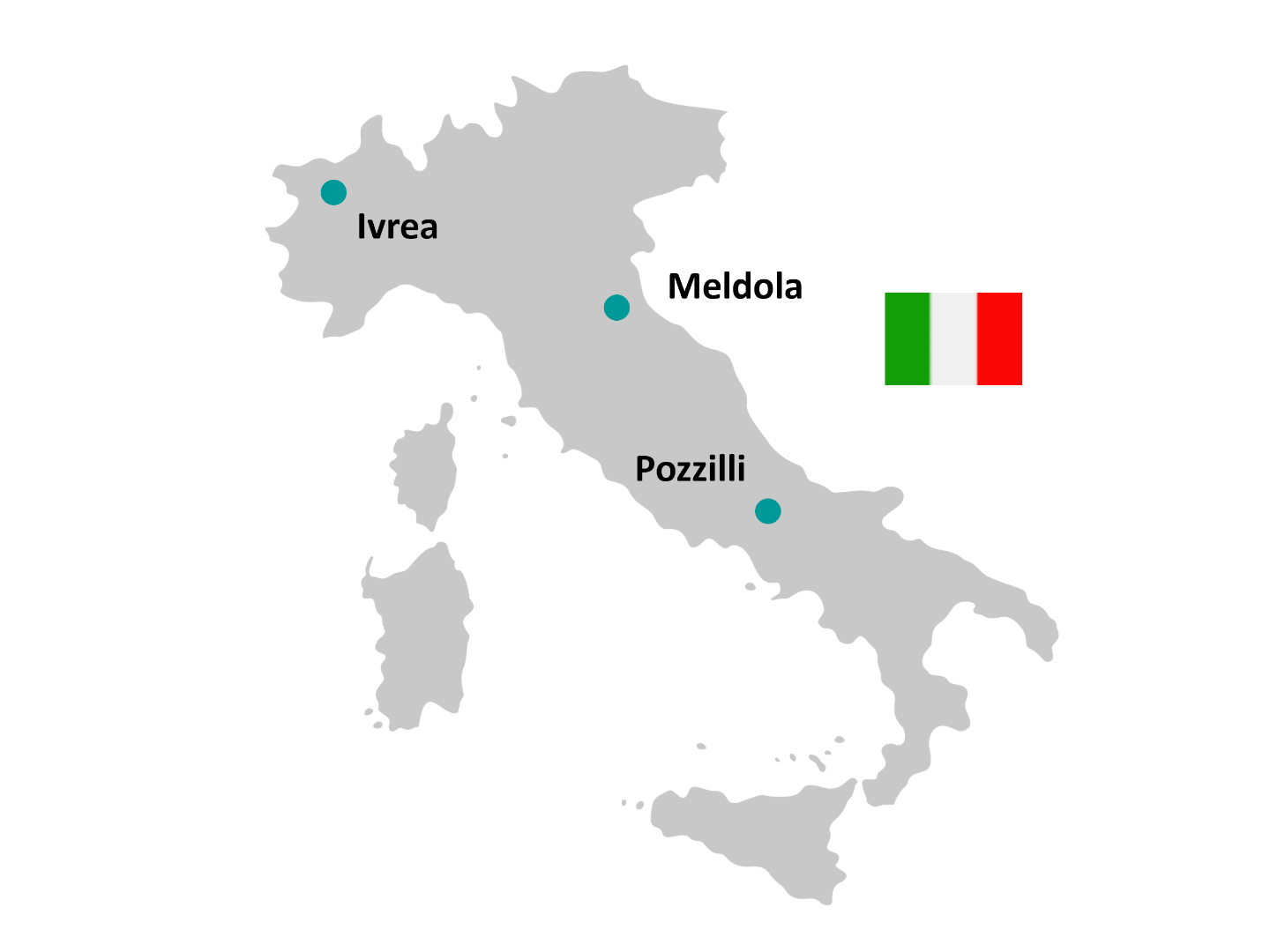
Ivrea is currently under construction Available PET radiopharmaceuticals
Additional PET radiopharmaceuticals are available, please contact your local pharmacy.
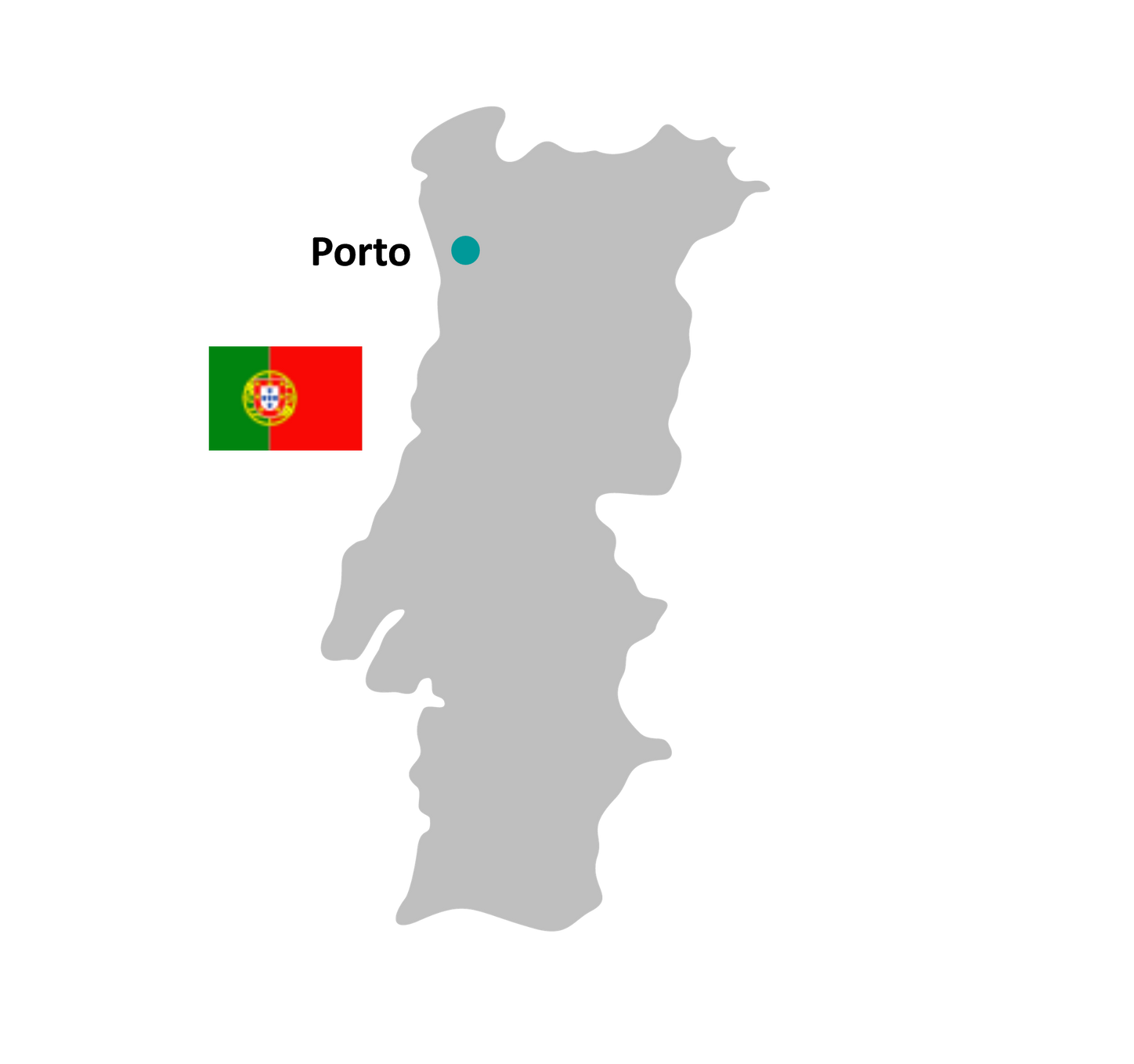
Available PET radiopharmaceuticals
Additional PET radiopharmaceuticals are available, please contact your local pharmacy.
Available PET radiopharmaceuticals
Gluscan
AAACholine
Salamanca is currently under construction Available PET radiopharmaceuticals
Additional PET radiopharmaceuticals are available, please contact your local pharmacy.
Available PET radiopharmaceuticals
MetaTrace FDG
Contact us
What is an adverse event?
Adverse events are any unintended or undesirable medical event experienced by a patient who has received a pharmaceutical product, which may or may not be directly caused by the treatment. An adverse event can include any unfavorable signs, symptoms, or medical conditions associated with the use of the product.
Why report an adverse event?
Reporting adverse events for Advanced Accelerator Applications Molecular Imaging products helps ensure the safety of our products and patients. This information also enables Siemens Healthineers to meet its reporting obligations to health authorities, requiring disclosure of adverse events—even when a link to the product isn’t established.
If you or the person you are reporting on behalf of are currently experiencing severe adverse events, contact your local doctor or healthcare professional before reporting.
Please contact Siemens Healthineers to report any adverse reactions related to our products.
When submitting in an e-mail format please provide the following information in your email to ensure it is handled in a manner consistent with the applicable local laws:
- Name of the drug:
- Description of when the patient started the medication, including dosing strength and frequency. Any action taken with the medication (e.g. reduced dose, stopped) and impact of that action on the adverse event.
- The adverse event associated with the drug:
- Symptoms evolve over time. Description of any treatment needed for the adverse event.
- Name of healthcare provider who provided treatment and contact information.
- Your name (optional):
- Please note that your personal identifying information (e.g. name, email address) will not be shared with the health authorities. Siemens Healthineers or its agents may contact you for further information about the adverse event. If you do not wish to be contacted, you should indicate this in your email.
- Information about the person who experienced the adverse event:
- Any other medical conditions currently ongoing. Medications currently taken (name, dosing strength, frequency, and when medication was started). Gender and age of the person taking medication (optional).
How to report adverse events?
By sending an email to the provided email address, I explicitly consent to the individual disclosure of my Personal Data for the above-mentioned purpose. I understand that this action confirms my voluntary consent.
To report the adverse events electronically please click below:
- Name of the drug:
Quality complaints
Advanced Accelerator Applications Molecular Imaging, a Siemens Healthineers Company, at 20 rue Diesel 01630 - Saint-Genis-Pouilly France, and its Affiliates (hereinafter collectively referred to as "Advanced Accelerator Applications Molecular Imaging, a Siemens Healthineers Company") is requesting the right to collect your personal data and to process, disclose, release and/or distribute such personal data for the purpose set out below. “Affiliate(s)” shall mean any entity which directly or indirectly controls, is controlled by or is under common control of Advanced Accelerator Applications Molecular Imaging, a Siemens Healthineers Company.
Advanced Accelerator Applications Molecular Imaging collects the following personal information (“Personal Data”):
- Name
- Contact information
- Profession
- Specialty
Advanced Accelerator Applications Molecular Imaging uses this information as follows:
- Purpose of processing: Advanced Accelerator Applications Molecular Imaging will process and document your above mentioned Personal Data in order to Respond to your Medical Information Request.
- Intra-group, cross-border transfer and data processing: The above mentioned data processing may be carried out by Advanced Accelerator Applications Molecular Imaging or by domestic or foreign Advanced Accelerator Applications Molecular Imaging Affiliates within or outside the European Union.
- Retention: Irrespective of the fact that your data was disclosed, your personal data will be kept for the duration required by regulatory requirements, and not less than the time the product/device you have received information about is available to the public. Your data will not be shared with any other group, nor used for any other purpose without your prior written consent. This does not affect statutory duties to preserve records beyond the stated retention period.
Advanced Accelerator Applications Molecular Imaging protects your Personal Data as follows:
Advanced Accelerator Applications Molecular Imaging has implemented appropriate technical and organizational measures to provide an adequate level of security and confidentiality to your Personal Data.
These measures take into account:
- the state of the art of the technology;
- the costs of its implementation;
- the nature of the data; and
- the risk of the processing.
The purpose thereof is to protect it against accidental or unlawful destruction or alteration, accidental loss, unauthorized disclosure or access and against other unlawful forms of processing.
Regarding intra-group cross-border transfer and data processing, Advanced Accelerator Applications Molecular Imaging, a Siemens Healthineers Company, has adopted Binding Corporate Rules, ensuring adequate safeguards are in place for intra-group cross-border transfers of your Personal Data.
Moreover, when handling your Personal Data, Advanced Accelerator Applications Molecular Imaging:
- only collects and processes personal data which is adequate, relevant and not excessive, as required to meet the above purposes; and
- ensures that your Personal Data remains up to date and accurate.
For the latter, Advanced Accelerator Applications Molecular Imaging may request you to confirm the Personal Data we hold about you. You are also invited to spontaneously inform us (by emailing
dataprivacy.func@siemens-healthineers.com) whenever there is a change in your personal circumstances, so Advanced Accelerator Applications Molecular Imaging can ensure your Personal Data is kept up-to-date.Your rights and how can you exercise them
You may exercise the following rights under the conditions and within the limits set forth in the law:
- the right to withdraw your consent at any time with effect for the future, without affecting the lawfulness of the processing before such withdrawal and without this resulting in any disadvantages for you;
- the right to access your Personal Data as processed by Advanced Accelerator Applications Molecular Imaging and, if you believe that any information relating to you is incorrect, obsolete or incomplete, to request its correction or updating;
- the right to request the erasure of your Personal Data or the restriction thereof to specific categories of processing;
- the right to request its portability, i.e. that the Personal Data you have provided to Siemens Healthineers be returned to you or transferred to the person of your choice, in a structured, commonly used and machine-readable format without hindrance from us and subject to your confidentiality obligations.
If you have a question or want to exercise the above rights, you may do so by sending an email to
dataprivacy.func@siemens-healthineers.comIf you are not satisfied with how Advanced Accelerator Applications Molecular Imaging processes your Personal Data, please address your request to our Data Privacy Team at dataprivacy.func@siemens-healthineers.com, who will investigate your concern.
In any case, you also have the right to file a complaint with the competent data protection authorities, in addition to your rights above.
Consent
By sending an email to the provided email address, I explicitly consent to the individual disclosure of my Personal Data for the above-mentioned purpose. I understand that this action confirms my voluntary consent.
Medical information
If you have questions regarding medical information such as safety, efficacy, dosing, and administration of our products, please click here for prescribing information
Molecular Imaging Medical Information Contact Form
Advanced Accelerator Applications Molecular Imaging, a Siemens Healthineers Company, with offices at 20 rue Diesel 01630 - Saint-Genis-Pouilly France, and its Affiliates (hereinafter collectively referred to as "Advanced Accelerator Applications Molecular Imaging, a Siemens Healthineers company") is requesting the right to collect your personal data and to process, disclose, release and/or distribute such personal data for the purpose set out below. “Affiliate(s)” shall mean any entity which directly or indirectly controls, is controlled by, or is under common control of Siemens Healthineers.
Siemens Healthineers collects the following personal information (“Personal Data”)- Name
- Contact information
- Profession
- Specialty
Advanced Accelerator Applications MI uses this information as follows:
- Purpose of processing: Advanced Accelerator Applications Molecular Imaging will process and document your above mentioned Personal Data in order to Respond to your Medical Information Request.
- Intra-group, cross-border transfer and data processing: The above mentioned data processing may be carried out by Advanced Accelerator Applications MI or by domestic or foreign Advanced Accelerator Applications Molecular Imaging Affiliates within or outside the European Union.
- Retention: Irrespective of the fact that your data was disclosed, your personal data will be kept for the duration required by regulatory requirements, and not less than the time the product/device you have received information about is available to the public. Your data will not be shared with any other group, nor used for any other purpose without your prior written consent. This does not affect statutory duties to preserve records beyond the stated retention period.
Advanced Accelerator Applications Molecular Imaging protects your Personal Data as follows:
Advanced Accelerator Applications Molecular Imaging has implemented appropriate technical and organizational measures to provide an adequate level of security and confidentiality to your Personal Data.
These measures take into account:- the state of the art of the technology;
- the costs of its implementation;
- the nature of the data; and
- the risk of the processing.
The purpose thereof is to protect it against accidental or unlawful destruction or alteration, accidental loss, unauthorized disclosure or access and against other unlawful forms of processing.
Regarding intra-group cross-border transfer and data processing, Advanced Accelerator Applications Molecular Imaging, as a member of Siemens Healthineers, has adopted Binding Corporate Rules, ensuring adequate safeguards are in place for intra-group cross-border transfers of your Personal Data.
Moreover, when handling your Personal Data, Advanced Accelerator Applications Molecular Imaging:- only collects and processes personal data which is adequate, relevant and not excessive, as required to meet the above purposes; and
- ensures that your Personal Data remains up to date and accurate.
For the latter, Advanced Accelerator Applications Molecular Imaging may request you to confirm the Personal Data we hold about you. You are also invited to spontaneously inform us by emailing
dataprivacy.func@siemens-healthineers.com whenever there is a change in your personal circumstances, so Advanced Accelerator Applications Molecular Imaging can ensure your Personal Data is kept up-to-date.Your rights and how can you exercise them
You may exercise the following rights under the conditions and within the limits set forth in the law:- the right to withdraw your consent at any time with effect for the future, without affecting the lawfulness of the processing before such withdrawal and without this resulting in any disadvantages for you;
- the right to access your Personal Data as processed by Advanced Accelerator Applications Molecular Imaging and, if you believe that any information relating to you is incorrect, obsolete or incomplete, to request its correction or updating;
- the right to request the erasure of your Personal Data or the restriction thereof to specific categories of processing;
- the right to request its portability, i.e. that the Personal Data you have provided to Siemens Healthineers be returned to you or transferred to the person of your choice, in a structured, commonly used and machine-readable format without hindrance from us and subject to your confidentiality obligations.
If you have a question or want to exercise the above rights, you may do so by sending an email to
dataprivacy.func@siemens-healthineers.comIf you are not satisfied with how Advanced Accelerator Applications Molecular Imaging processes your Personal Data, please address your request to our Data Privacy Team at
dataprivacy.func@siemens-healthineers.com, who will investigate your concern.
In any case, you also have the right to file a complaint with the competent data protection authorities, in addition to your rights above.By completing the form below, I explicitly consent to the individual disclosure of my personal data for the purpose as mentioned above. I understand that this action confirms my voluntary consent.
Did this information help you?
Thank you.
Would you like to provide detailed feedback?
125 / 125
