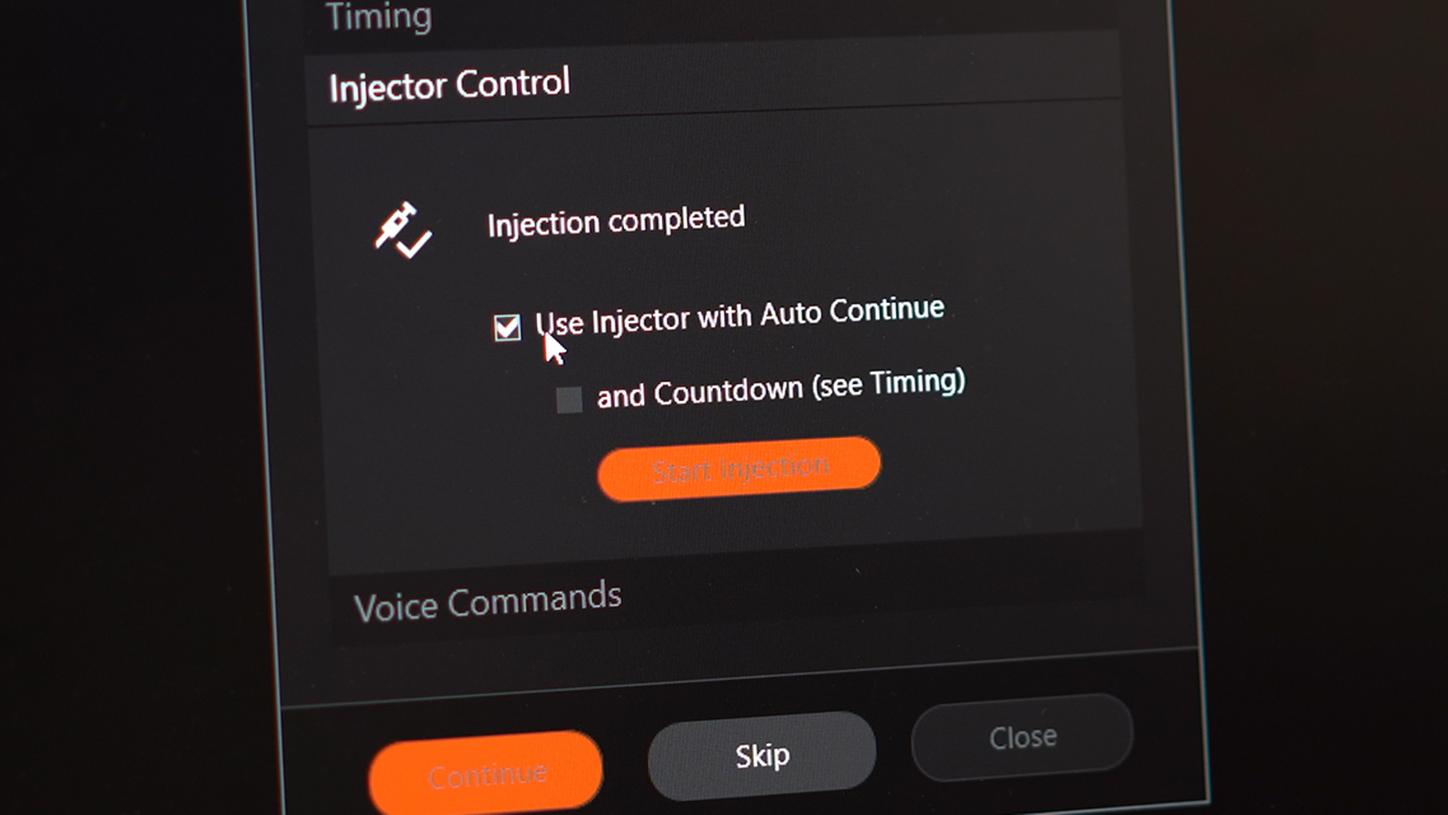
MR-Injector coupling via Imaging System Interface (ISI)1Simplified & synchronized MR contrast injection & scanning
MR-Injector coupling via ISI is a joint development by Bayer and Siemens Healthineers. It substantially simplifies synchronized contrast injection and scanning by guiding the user through an intuitive workflow with clicks and interactions reduced to the essence. Instead of multiple, time-critical interactions on different consoles (MR host, Injector console, Intercom with Patient), everything is controlled with a few clicks from the MR console. By also supporting remote operation scenarios, ISI may help to provide advanced contrast-enhanced imaging services in remote areas – instead of enforcing patients to travel to distant, centralized expert centers.
1
The products/features (here mentioned) are not commercially available in some countries. Due to regulatory reasons their future availability cannot be guaranteed. Please contact your local Siemens organization for further details.
The information shown herein refers to products of 3rd party manufacturer’s and thus are in their regulatory responsibility. Please contact the 3rd party manufacturer for further information.
The information shown herein refers to products of 3rd party manufacturer’s and thus are in their regulatory responsibility. Please contact the 3rd party manufacturer for further information.

