History
A 75-year-old male patient, with a history of esophagectomy 10 years ago, presented to the emergency department with an acute onset of epigastric pain radiating to the left upper quadrant, chest pain, obstipation and numerous episodes of vomiting. On physical examination, the patient was diaphoretic and tachycardic, showing guarding in the upper abdomen. Laboratory results revealed a lactate level of 3.0 mmol/L, an estimated GFR of > 60 and a WBC of 11. A CT examination of the abdomen and pelvis with contrast was immediately performed for evaluation.
Diagnosis
CT images revealed multiple dilated loops of small bowel herniating into the thoracic cavity through a defect in the left hemidiaphragm. A thin rim of hyperdensity within the wall of the herniated bowel loops was seen in the images reconstructed conventionally, indeterminate of mucosal enhancement (suggesting no ischemia) or hemorrhage within the submucosa (suggestive of infarction). In the assessment of spectral imaging data, a complete lack of enhancement within the herniated loops of bowel was shown in the iodine maps, compatible with infarction. The hyperdensity in the bowel wall was present in the virtual non-contrast (VNC) images, suggestive of hemorrhage in the submucosa. With this added spectral information, a diagnosis of small bowel infarction was made, and the patient was taken rapidly to surgery for small bowel resection. Pathological evaluation on the resected bowel confirmed complete bowel necrosis with hemorrhagic blood products throughout all bowel layers and sloughing of the mucosa. The patient recovered with good outcome.

Courtesy of Department of Radiology and Radiological Sciences, The Medical University of South Carolina, Charleston, SC, USA
Fig. 1: A coronal view of a VRT image shows herniated loops of small bowel (arrow) into the left thoracic cavity.
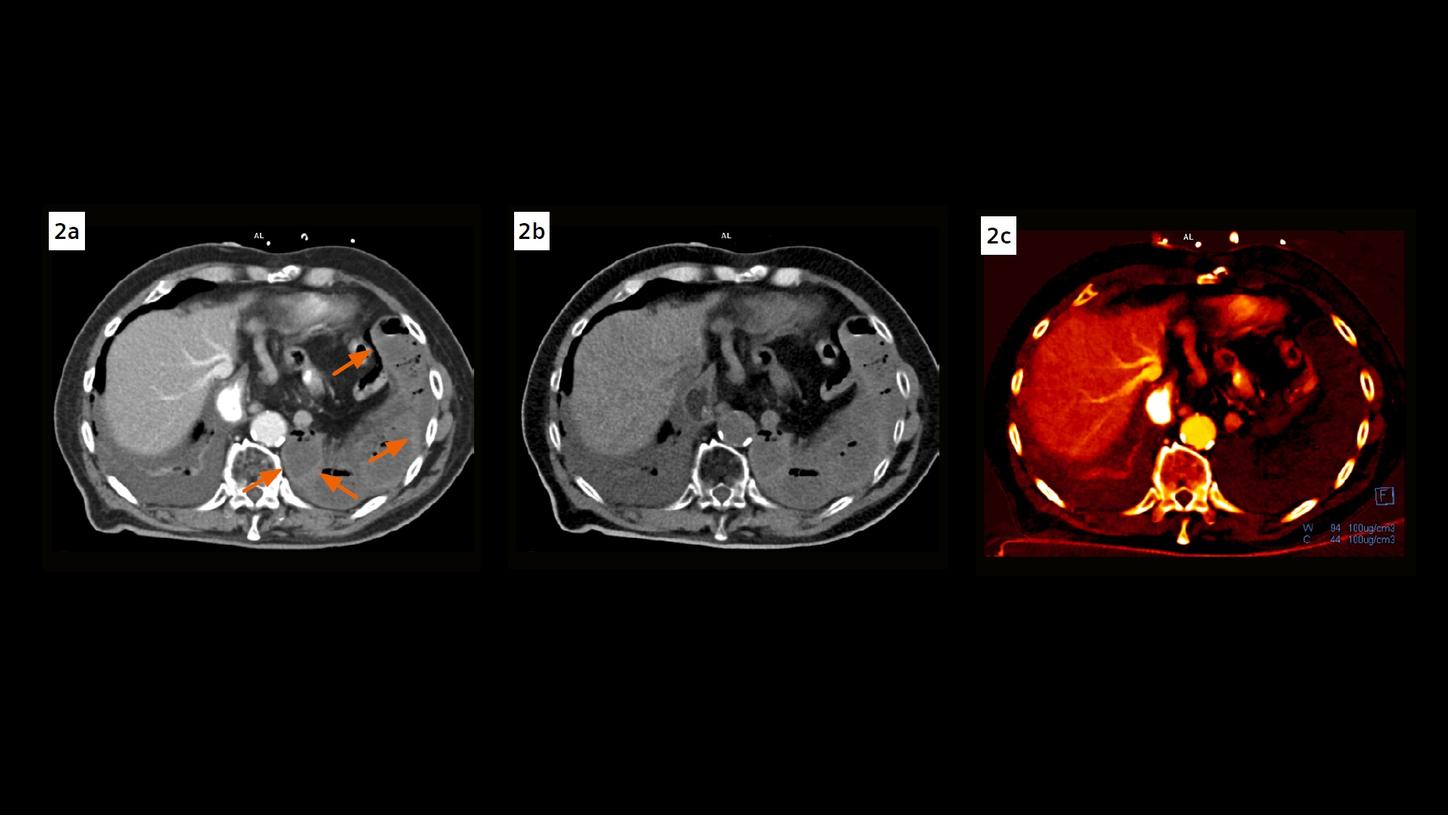
Fig. 2: A contrast enhanced axial image (Fig. 2a) shows a thin rim of hyperdensity within the wall of the herniated bowel loops (arrows), which are also seen in the VNC image (Fig. 2b), but not seen in the iodine map (Fig. 2c), suggesting submucosal hemorrhage, compatible with infarction.
Comments
Bowel infarction refers to vascular compromise of portions of the bowel. It can occur secondarily to an acquired diaphragmatic hernia in patients with prior surgery due to altered anatomy, such as this case. The initial clinical presentations including physical exam and laboratory abnormalities are usually nonspecific. However, the progress from an initial necrosis of the bowel wall to a final sepsis and multiorgan failure can go rapidly. Treatment is often surgical and imaging is relied upon for rapid diagnosis. In recent years, CT has largely replaced conventional angiography due to rapid access and accuracy. On CT imaging, ischemic segments of bowel are often found to have a different density and enhancement compared to nonischemic segments. However, the subtlety of positive image findings may go underappreciated and can lead to unnecessary delay or even misdiagnosis. In this case, the thin rim of hyperdensity demonstrated in the bowel wall could have been interpreted as normal mucosal enhancement in viable bowel. However, its presence in the VNC images and absence in the iodine maps characterize an infarction. The added spectral information improves reader confidence, and then, taking into account the clinical and laboratory findings, a rapid diagnosis can be made which is essential for the patient to receive prompt treatment. The CT scan was performed with NAEOTOM Alpha, a newly developed CT scanner with photon-counting detectors. It provides energy-resolved CT data with a high spatial resolution, without electronic noise. [1] The availability of spectral imaging data in routine scans is one of the major improvements in workflow. Conventional CT image reading can be freely switched to spectral image reading with just one mouse click, making the assessment of diagnostic information more efficient and fast. As presented in this case, spectral CT may overcome the traditional shortcomings of conventional CT in detecting bowel infarction by adding iodine-sensitive information, enabling the differentiation of hemorrhage from true contrast-material uptake, i.e., blood perfusion, with distinctive image findings.
Examination Protocol