- Home
- Healthcare IT
- Laboratory Diagnostics IT
- Atellica Diagnostics IT
- Atellica Data Manager
Atellica Data Manager
Laboratory data management software to standardize testing and streamline result management across your diagnostics network
Accelerate and standardize result reviews. Reduce errors. Simplify and strengthen QC management. Enhance clinical decisions. Support compliance requirements. Enable audit-readiness. More open and scalable than ever before, Atellica® Data Manager is laboratory data management software designed to increase visibility and centralize management across a broad spectrum of multidisciplinary instruments and sites.
Features & Benefits
Get the functions you need most!
It’s never been easier to digitalize best practices that have proven effective in thousands of labs around the world using laboratory data management software that has evolved over more than 22 years!
Quick-click navigation to:
- Rules-based Standardized Testing
- Realtime Workflow Dashboard
- Integrated QC Management
- Simplified Review and Edit
- Built-in Result Autovalidation
- Robust Traceability, Audit Trails, and Reporting



Extensive Rules Library
Consider how you can streamline workflows by applying data-driven rules and algorithms for calculated tests, pre- and post-analytics, rerun and reflex testing, and clinical decision support.
Simplify operations with an at-a-glance view of your lab’s workload. Review and edit results with screens designed for quick-click navigation and action. Use IT to amplify the power of lab automation.

Amplify the Power of Automation
We offer scalable automation for labs of all sizes. Atellica Data Manager supports rule-based testing and result management to increase the productivity of our analyzer-integrated, task-targeted, and/or total laboratory automation offerings.

Real time dashboard
- Operator-specific dashboards
- Panic Counters
- Shortcuts
- Support staffing flexibility
- Simplified training, etc.

Efficient Result Management
- Review by exception
- Everything you need on a single screen
- Multitasking made easy

Amplify the Power of Automation
We offer scalable automation for labs of all sizes. Atellica Data Manager supports rule-based testing and result management to increase the productivity of our analyzer-integrated, task-targeted, and/or total laboratory automation offerings.

Real time dashboard
- Operator-specific dashboards
- Panic Counters
- Shortcuts
- Support staffing flexibility
- Simplified training, etc.

Efficient Result Management
- Review by exception
- Everything you need on a single screen
- Multitasking made easy

Amplify the Power of Automation
We offer scalable automation for labs of all sizes. Atellica Data Manager supports rule-based testing and result management to increase the productivity of our analyzer-integrated, task-targeted, and/or total laboratory automation offerings.




Configurable Dashboard
Result Management
Quality Control
Powerful Rules &
Clinical Decision Support Capabilities
Sample Query Browser
Put your data to work
It can be time consuming, cumbersome, and expensive to work with individual analyzer software, to consolidate data in the laboratory information system, or to deal with functional gaps in immature middleware. Atellica Data Manager helps overcome these issues to deliver the results you care about!

Focus your staff on what matters most.
Focus your staff on what matters most.
- Help avoid information overload with modern, sleek, and simple interface.
- Tailor views, editing, and administrative privileges to each user’s needs.
- Spot problems easily with color codes, severity alerts, and customized dashboard counters.
- Access information and take action quickly with quick-click navigation.
- Add comments and manage single or multiple samples simultaneously.
- Simplify staff access to the application with optional single sign-on to the hospital network via Active Directory.

Operationalize your data using powerful rules
Operationalize your data using powerful rules.
- Digitalize best practices for QC testing and result management. Use data-driven rules and built-in functions to identify and investigate nonconforming issues.
- Quickly document corrective actions to help satisfy compliance and audit requirements.
- Automate workflows for sample processing and reflex and repeat testing.
- Implement rules to drive algorithm-based infectious-disease testing and facilitate ISLH hematology guidelines.
- Coordinate pretest dilution processing, increase autovalidation rates, and more.
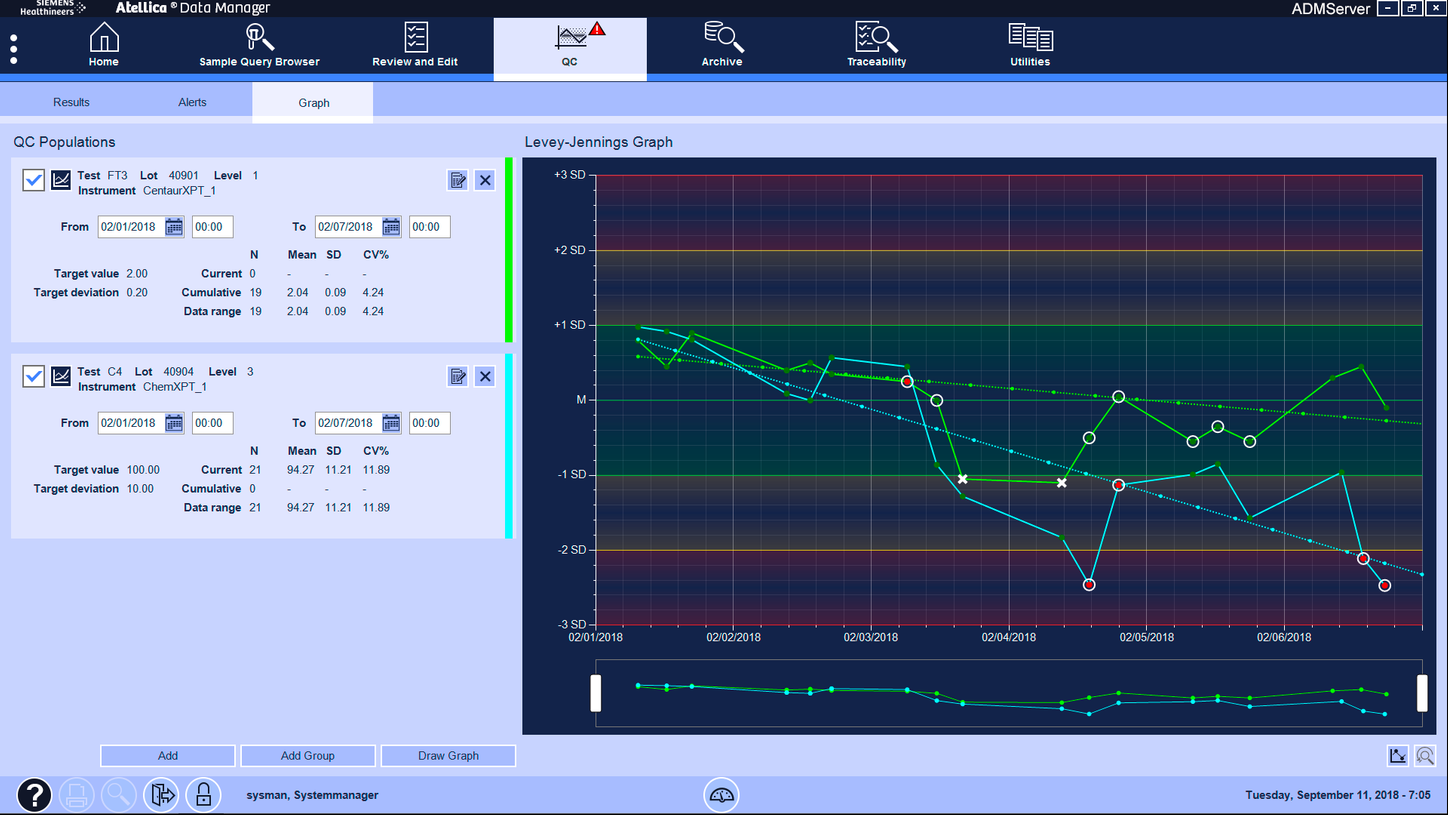
Simplify daily management and monthly reviews.
Simplify daily management and monthly reviews.
- Automatically identify QC issues, hold reporting of corresponding patient results, and prevent subsequent testing.
- Enable staff to easily identify and reprocess affected tests with just a few clicks.
- Monitor QC using lab-defined, assay-specific Westgard rules.
- Use patient-based moving averages to proactively identify trends—across lots, instruments, and assays.
- Plot and compare multiple QC lots, levels, and instruments.
Support multiple sites with unlimited instrument connections, unlimited users, and unlimited concurrent sessions*.
*Performance claims are validated with 125 connected instruments, a throughput of 150,000 tests per hour, and 75 concurrent user sessions.
Sharpen your clinical focus.
- Centralize management across multidisciplinary instruments, labs, and LISs with open, scalable software.
- Provide individual uses with easy access to details needed for informed, accurate decisions.
- Evaluate and accommodate patient-specific scenarios using patient demographics along with absolute and percentage-based delta checks.
- Support staff adherence to lab protocols by providing scenario-specific information and automating tasks.
Lighten workloads.
- Evaluate and verify patient results automatically based on predetermined criteria established by your lab for normal ranges, delta checks, quality control, and instrument flag severities.
- Review only those results that require further action before reporting.
- Add objectivity and consistency to the result review process.
- Help technologists not to miss abnormal results and focus their attention where it is needed most. Leverage sample-to-consumable and consumable-to-sample traceability.
Technical Specifications
Used by some of the world’s largest laboratories, the robust performance of Atellica Data Manager evolved over many years. The software can be easily configured to support unlimited instrument connections, users‡, and concurrent sessions‡. Connect and manage multiple labs, LISs, automation systems, and multidisciplinary instruments using a highly scalable, virtual, and open software solution with access to a growing library of more than 700 device drivers supporting approximately 1500 analyzer models and IT solutions from these and other manufacturers:
- A. Menarini
- Abbott
- Adaltys
- Alifax
- Beckman Coulter
- Benson Viscometers Ltd.
- bioMérieux
- Bio-Rad
- CellaVision
- COPAN
- Diagnostica Stago
- DiaSorin
- Diatron
- DIESSE Diagnostica Senese
- Dynex Technologies
- Eiken Chemical Co.
- Fujirebio
- Grifols
- Helena Laboratories
- Hitachi
- Hologic
- IDS
- Inova Diagnostics
- Instrument Laboratories
- Intellitec Healthcare IT Solutions GmbH
- JEOL
- MIKROGEN Diagnostik
- NGNY Devices S.L.
- Ortho Clinical Diagnostics
- Roche
- RR Mechatronics
- Sebia
- Seegene, Inc.
- SERION Diagnostics
- Siemens Healthineers
- Snibe Diagnostic
- Sysmex
- TECAN
- Thermo Fisher Scientific
- Tosoh Bioscience
- Trinity Biotech
- Waters Corporation
Click here to see a current list of instrument interfaces that are supported by Atellica Data Manager. Availability of products varies by country. Ask your local Siemens Healthineers representative for a list of instruments available in your local market.
Testimonials
Proven by labs around the world
We have decades of experience implementing advanced laboratory data management systems and digitalizing best practices in open, multidisciplinary labs.
Since 1998, more than 2000* labs in 68 countries have trusted successive generations of this robust software to optimize clinical operations and increase workforce productivity.
"We love Atellica Data Manager. It includes all the functionality, power, and reliability of CentraLink® [system] but with very modern and easy-to-use software."

Dr. Gustavo Dip
Technical Director, Lab Turner
"Atellica Data Manager has provided us with simplicity, helped with quality, made us more efficient, and enabled us to meet our customer needs in a way that we have never been able to before."

Susan Dawson
Lab Director, Swedish Hospital
"Atellica Data Manager is very powerful and flexible. We’ve set up customized shortcuts on our home screen that enable our lab techs to work faster. [It] can reduce our TAT and lean our workflow."

Ting Hu
Automation Team Leader, Zhongshan People’s Hospital
"The new home screen really gives an immediate overview of where your attention is needed and provides immediate access to quick action."

Dr. Gustavo Dip
Technical Director, Lab Turner
"The improvement in QC capabilities and tracking allows users to review QC with just one click."

Susan Dawson
Lab Director, Swedish Hospital
Education & Training
Consider what you can achieve
Large and small hospital and reference laboratories are reaping the benefits of advanced data management.
Webinars
Did this information help you?
Thank you.
Customer list as of July 2020 on file.
Performance claims are validated with 125 connected instruments and a throughput of 150,000 tests per hour and 50 concurrent user sessions.
Interface and driver library as of September 2018.
The outcomes achieved by the Siemens Healthineers customer described here were achieved in the customer’s unique setting. Since there is no typical hospital or laboratory, and many variables exist (e.g., hospital or laboratory size, case mix, level of IT adoption), there can be no guarantee that others will achieve the same results.
Product availability may vary from country to country and is subject to varying regulatory requirements. Please contact your local representative for availability.





