Scalable COVID-19 antigen testing in 15 minutes
Simple procedure, reliable results
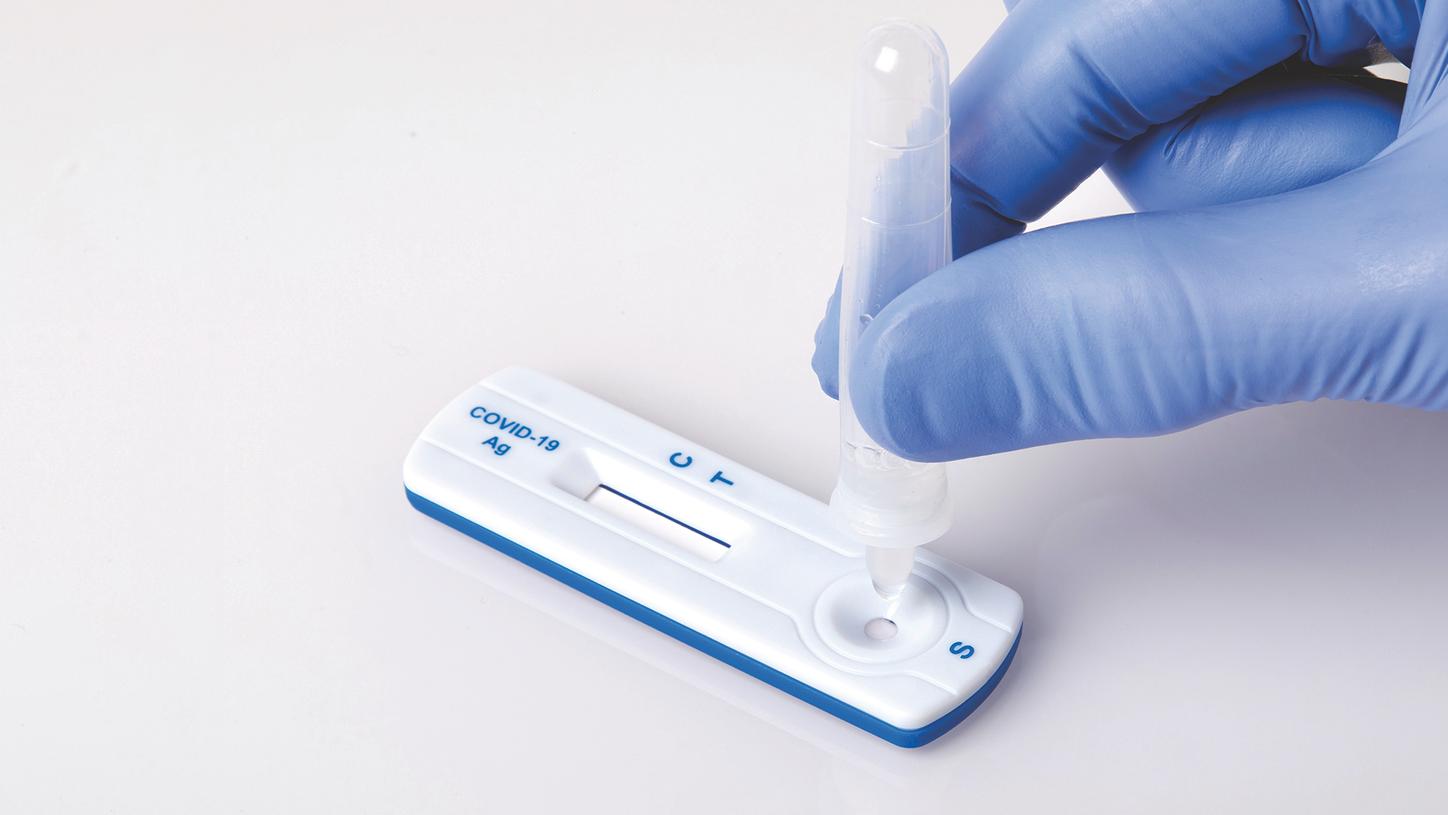
The lateral flow CLINITEST Rapid COVID-19 Antigen Test, distributed by Siemens Healthineers, is easy to administer by any trained professional in virtually any setting, without special equipment. When used as part of a comprehensive strategy, the CLINITEST Rapid COVID-19 Antigen Test can help communities stay ahead of the spread of COVID-19, even in the face of emerging variants.
Download the IFU
Official instructions for use for the CLINITEST COVID-19 Rapid Antigen Test:
Accessible testing, where you need it most
The CLINITEST Rapid COVID-19 Antigen Test can be used to test symptomatic or asymptomatic patients in a wide variety of settings.




