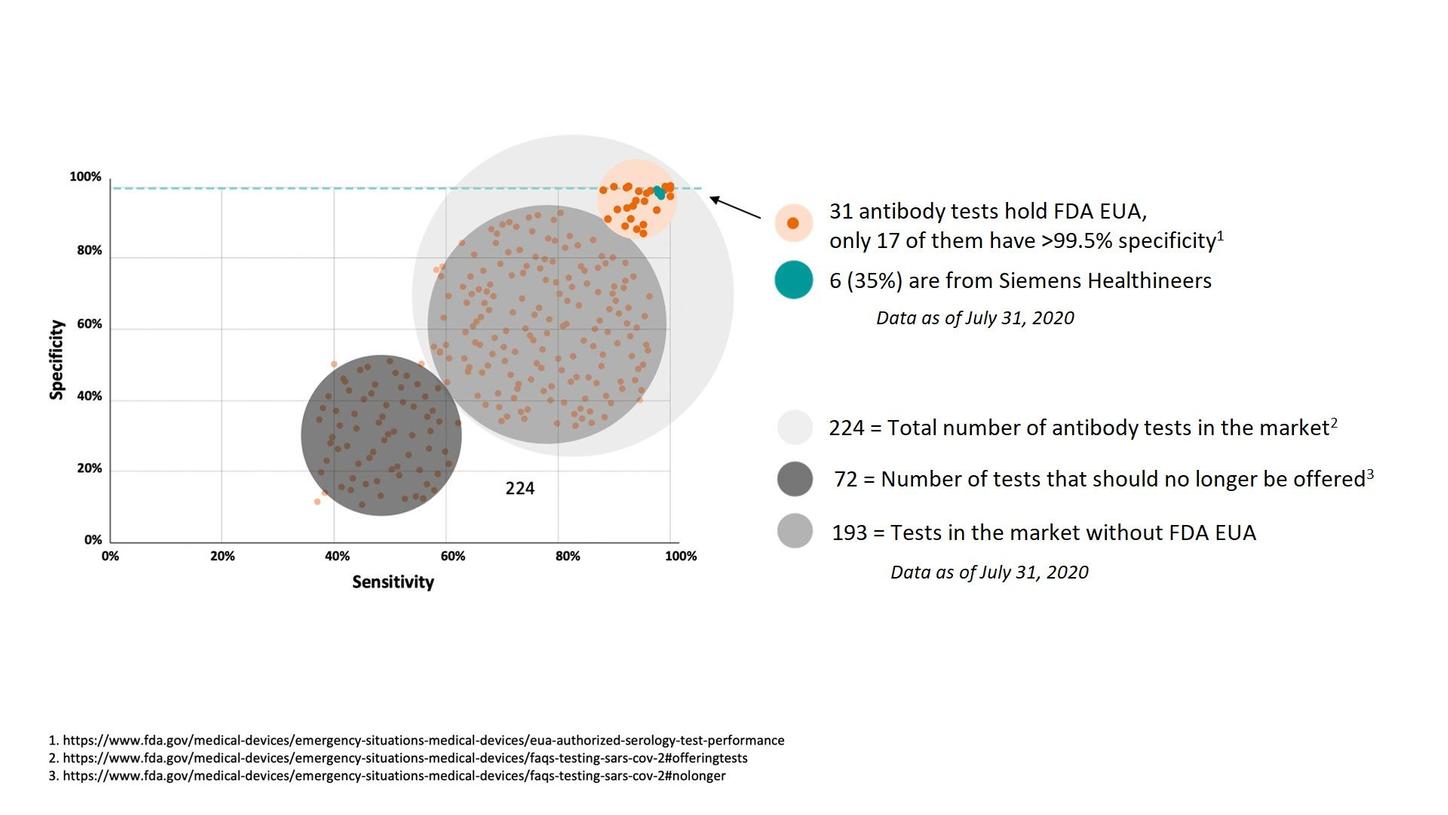
Sensitivity vs Specificity
- Test sensitivity indicates the ability of the test to correctly identify patients that have the disease. A test’s sensitivity is also known as the true positive rate. If a diagnostic test correctly identified 100% of all positive results, it would be as sensitive as possible.
- Test specificity indicates the ability of the test to correctly identify patients that do not have the disease. If a test correctly identifies all people without the disease as negative, it would be as specific as possible.
Specificity in COVID-19 testing
There are numerous tests that claim to detect antibodies to the SARS-CoV-2 virus; only a few are highly accurate:
How do highly specific antibody tests support good performance, even with low disease prevalence?
What happens when the population is tested with an assay that has higher specificity of 99.8%?
What happens when the specificity is reduced to 96%?
The model above assumes assay sensitivity of 100%